Revista Veterinaria. 2026; 37(1)
https://doi.org/10.30972/vet.3719047
Artículo Original




1AER INTA Goya. Av. José Jacinto Rolón 736 (W3450), Goya.
2EEA INTA Mercedes, Juan Pujol al este sin número, (W3470) Mercedes, Corrientes.
3CONICET. Godoy Cruz 2290 (C1425FQB) CABA –Argentina. 4Moredun Research Institute, Pentland Science Park, EH26 0PZ, Penicuik, UK.
Recibido: 15 mayo 2025 / Aceptado: 16 octubre 2025
Abstract
The aim of the present study was to determine the association between serological status and the causes of abortion related to bacterial and protozoan infections in three livestock farms with low reproductive performance in Corrientes province, Argentina. During 2020-2021, preputial scraping of bulls, pregnancy diagnosis by transrectal ultrasound in cows, and collection of aborted fetuses were performed. Blood samples from pregnant, non-pregnant and aborted cows were analyzed for Leptospira spp., Neospora caninum and Brucella abortus antibodies. In addition, cervicovaginal mucus (CVM) samples were collected from aborted cows for culture. Five percent of the bulls were positive for Tritrichomonas foetus and 7% for Campylobacter fetus (locality 1). Among 546 sera analysed, 1.8% (10/546; 95% CI 1-3) were positive for Leptospira spp., showing a significant association between high seroprevalence and non-pregnant females (p = 0.0174). For N. caninum, the seroprevalence was 49.1% (268/546 95% CI 45-54), showing a statistically significant association between seropositivity and pregnant females (p = 0.0151). Of 14 aborted females monitored, 42.8% became seropositive to N. caninum, suggesting horizontal transmission, a finding of notable importance. All animals were seronegative for B. abortus. Furthermore, T. foetus (3/61) and C. fetus (3/61) were isolated from 9.8% (6/61) of CVM samples, and infectious agents were identified as the cause of abortion in 42.9% (3/7) of the fetuses. This study provides insight into infectious causes of reproductive losses in extensive cattle herds in the Argentine Mesopotamia, highlighting the importance of venereal disease in bulls and the need for sampling of aborted cows and fetuses to identify causes of reproductive failure.
Key words: abortion, reproductive losses, cattle, cervicovaginal mucus.
Resumen. El objetivo del presente estudio fue determinar la asociación entre el estado serológico y las causas de abortos relacionadas a infecciones bacterianas y protozoarias en tres establecimientos ganaderos con bajo desempeño reproductivo en la provincia de Corrientes, Argentina. Durante 2020-2021, se realizó raspado prepucial de toros, diagnóstico de gestación por ecografía transrectal en vacas y recolección de fetos. Se tomaron muestras de sangre de vacas gestantes, no gestantes y abortadas para el diagnóstico serológico de Leptospira spp., Neospora caninum y Brucella abortus. Además, se tomó una muestra de moco cervicovaginal (CVM) para cultivo en vacas abortadas. En el 5% de los toros fueron positivos para Tritrichomonas foetus y en el 7% para Campylobacter fetus (localidad 1). En el 1,8% (10/546; IC95% 1-3) de 546 sueros analizados fueron positivos para Leptospira spp., observándose una asociación positiva entre la alta seroprevalencia y las hembras no gestantes (p = 0,0174). Además, para N. caninum, la seroprevalencia fue del 49,1% (268/546 IC95% 45-54), mostrando una asociación estadísticamente significativa entre la serología positiva a N. caninum y las hembras preñadas (p = 0,0151). De 14 hembras abortadas en el seguimiento, el 42,8% se volvió seropositiva a N. caninum, lo que podría indicar una infección por transmisión horizontal, siendo este hallazgo de gran importancia. Todos los animales fueron seronegativos para B. abortus. Además, T. foetus (3/61) y C. fetus (3/61) se aislaron en el 9,8% (6/61) del CVM. Solo en el 42,9% (3/7) de los fetos, como agentes infecciosos se identificaron como causa del aborto. El presente estudio muestra información sobre la causa infecciosa de pérdidas reproductivas en rebaños extensivos de la Mesopotamia argentina, confirmando además la importancia de la enfermedad venérea en toros y el muestreo de vacas y fetos abortados para identificar la causa de la pérdida.
Palabras clave: aborto, pérdidas reproductivas, bovino, mucus cervicovaginal.
Reproductive diseases cause infertility and abortions in cattle, causing significant economic losses that frequently go unnoticed (Reichel et al. 2018, Baumgartner 2021). Various causes may influence the viability of the fetus, and therefore, sporadic and epidemic abortions frequently occur without previous clinical signs (Seid et al. 2021). Once fetal death has occurred, the fetus can be expelled or remain retained in the uterus, suffering autolysis, which makes it difficult to identify lesions and infectious agents (Reichel et al. 2018, Baumgartner 2021, Hecker et al. 2023).
Both infectious and non-infectious causes can be associated with these losses. Previous studies have shown that Neospora caninum infection is a major risk factor for reproductive losses in farmed cattle and is widely distributed (Moore et al. 2002). As for abortions caused by bacteria such as Leptospira spp., Campylobacter spp. and Brucella spp. are usually overlooked due to the lack of practical. These agents are implicated in cattle abortion during mid- to late-gestation posing zoonotic risks and economic loss.
These agents are known to be transmitted primarily through fecal-oral route, intrauterine, sexual or direct contact with infected material, resulting in neonatal death, abortion, urogenital system infections, and low productivity in ruminants (Givens and Marley 2008). The aim of the present study was to determine the association between serological status and the causes of abortion related to bacterial and protozoan infections in three livestock farms with low reproductive performance in the province of Corrientes, Argentina.
Bull analysis. During the period between 2020 and 2021, extensive Braford cattle herds (n = 3) located in the province of Corrientes, Argentina, showing pregnancy rates of around 75%, were monitored.
Three consecutive preputial scraping samples (PS) were collected from 1,053 bulls at 10-day intervals (livestock farms 1: 57 bulls (3%); livestock farms 2: 37 bulls (2%); livestock farms 3: 959 bulls (6%). The preputial area was trichotomized prior to sampling. Two scrapers were inserted into the preputial orifice, and thirty scraping movements were performed. The material was then placed into specific culture media for the detection of Tritrichomonas foetus by culture (Diamond 1983) and PCR (Felleisen 1997), and C. fetus by direct immunofluorescence (DIF) in 1% formol solution (Cipolla et al. 1994). Blood was also collected for B. abortus control.
Tracking pregnant, non-pregnant and aborted cows. Pregnant cows n = 375 were randomly selected (control group), which underwent 1 quarterly ultrasound and were bled once (time 0). Non-pregnant n:110 were bled once (time 0), and aborted females (fetuses were not found) n:61 had blood and cervicovaginal mucus (CVM) drawn between 7 and 30 days post-abortion. Cervico-vaginal mucus collection was performed using a Casso pipette, placing the mucus in specific transport media (AMIES) (4 °C, Diamond 37 °C, Cary-Blair 4 °C). The samples were preserved and analyzed in the laboratory within 24 hours after collection. From the aborted cows, 14 were from the control group flock, the remaining 47 belonged to the general herd and were not bled at time 0.
Blood samples will be used to determine the seroprevalence of Leptospira spp. with 1: 200 cut-off titer by microagglutination test-MAT (International Office of Epizootics [OIE], 2008), N. caninum with 1: 100 cut-off titer by indirect fluorescent antibody technique (IFAT) as previously described (Hecker et al. 2019) and B. abortus by buffer plate antigen test (BPA) and fluorescence polarisation assay (Nicola et al. 2019). Bacteriological culture (Holt et al. 1994) and diagnosis of T. foetus by culture (Diamond 1983) and PCR (Felleisen 1997) were performed on the CVM. For bacteriological culture, culture media were used plates with McConkey agar (MC, aerobiosis), Columbia blood agar with 7% bovine blood (ASC, in an atmosphere with 10% CO2), and Skirrow agar with 7% defibrinated equine blood (ASK, in an atmosphere created with gas tubes with 5% O2, 5-10% CO2 and 85-90% H2), incubating for 1, 2 and 7 days respectively, in an oven at 37 °C.
Necropsy and sampling of aborted fetuses. Aborted fetuses belonging to the general herd were analyzed by necropsy, using standard gross pathology procedures (Campero et al. 2003). Sterile samples of lung, spleen and abomasal content samples were collected to determine the presence of aerobic and microaerophilic bacteria by bacteriological culture (Holt et al. 1994). Fetal cavity fluids were collected and stored at −20 ºC until assessment of antibodies to N. caninum by IFAT. Additionally, fetal tissue samples (central neural system -CNS-, heart, lung, liver, tongue, forelimb and hind limb muscles) were fixed in 10% neutral buffered formalin for histopathological analysis as was previously described (Campero et al. 2003).
Established criteria for etiological diagnosis in fetuses. A diagnosis of congenital infection was made when isolation and/or IFAT results were positive. Furthermore, the cause of abortion was confirmed according to the following criteria: (i) presence of compatible histopathological lesions (ii) no other infectious agents diagnosed. Finally, a diagnosis was considered indeterminate when no causative agent could be detected by any of the laboratory tests.
Statistical analysis. Statistically significant differences in seroprevalence of Leptospira spp., B. abortus and N. caninum were determined. The chi-square test was used in order to evaluate possible associations. Descriptive analysis and expression of results were carried out based on total numbers and percentages of diagnosed cases. All statistical analyses were performed using the InfoStat app (Di Rienzo et al. 2020). A significance level of p˂0.05 was used.
After PS, 8.8% (5/57) of locality 1 of bulls were positive for T. foetus and 12.3% (7/57) of locality 1 for C. fetus. The rest of the localities did not present positive bulls. Of the 546 sera analyzed by MAT, 1.8% (10/546 CI95% 1-3) were positive for the Canicola, Pomona and Hardjo serovars. Of this total, 40% (4/10) were pregnant [Canicola (1/ 200), Pomona (1/400) and Hardjo (1/800) serovars], 60% (6/10) were non pregnant [serovars Canicola (1/400) and Hardjo (1/800) present] and the remaining 10% (1/10) were aborted (serovar Canicola 1/200 present). An association was found between the greater probability that non pregnant females have positive serology for Leptospira spp. in relation to pregnant females (p = 0.0174). However, no association was found with aborted females (p>0.05).
For N. caninum, a seroprevalence of 49.1% (268/546 CI95% 45-54) was found. Of this total, 71.6% (192/268) were pregnant females, 20.5% (55/268) were non-pregnant females, and 7.8% (21/268) were females that had aborted. A statistically significant association was found between positive serology and pregnant females (p = 0.0151). In contrast, an increase in N. caninum seropositivity of 42.8% (6/14) was observed in females that aborted (Table 1). No positive serology was found for B. abortus.
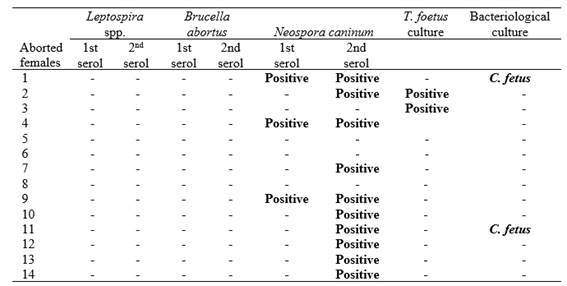
Abbreviations: T. foetus: Tritrichomonas foetus, C. fetus: Campylobacter fetus; -: Negative. serol: Serology
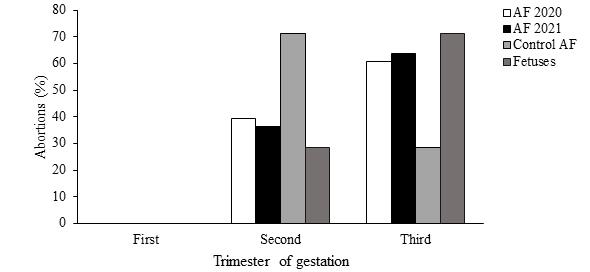
For venereal agents, 5.9% (3/61) for T. foetus (3/61) and 5.9% (3/61) for C. fetus were isolated and identified in CVM. The abortions of the control herd occurred mainly during the second trimester of gestation (Figure 1). When analysis was performed in the total aborted females (61) and seven fetuses (between 6 and 9 months) they were observed that abortions were distributed mainly in the third trimester of gestation (Figure 1). According to the established criteria in fetus, 42.9% (3/7) of the total losses were categorized as caused by infectious agents, being 2 caused by bacterial agents and 1 by N. caninum. In the fetuses analyzed, in those cases where the presence of the infectious agent was detected through the diagnostic techniques applied, histopathological lesions compatible with the infection caused by that agent were also observed. In the remaining 57.1% (4/7), the cause of death could not be established. Of the total number of fetuses analyzed, 2/7 presented antibodies against N. caninum, but in one of these two, no histological lesions compatible with the agent were observed, so their findings were rejected. In three of the four fetuses in which the agent was not identified, no histopathological lesions compatible with infectious processes were observed. In the remaining case, histological lesions suggestive of the involvement of an infectious agent in the etiology of abortion were identified. Regarding the distribution of abortions (Figure 1) in the first trimester, aborted females and fetuses were not observed.
Even though the area contains around 25% of the cows of Argentina, the soil characteristics and grazing systems result in a low density and extensive cattle production. This means that the livestock grasses in high extension paddocks are under minimal supervision, making the diagnosis of reproductive losses challenging. In the present study, the diagnosis of reproductive diseases was performed in bulls, cows, and fetal samples to prove them to be a feasible alternative for diagnosis. This study’s results establish the importance of bacterial and protozoal causes for reproductive losses in the bovine production systems of Corrientes in Argentina.
Regarding viral agents, they were not evaluated in the present study; however, indeterminate losses could have been associated with these agents. Also, although fetal histopathology can be valuable for identifying specific infectious agents, the main objective of this study was to highlight the relationship between serology and microbiological findings under the specific conditions of livestock production in northeastern Argentina. Under the local extensive production systems, where paddocks may have hundreds of hectares and livestock coexist with wildlife, the recovery of aborted fetuses is incidental and cannot be consistently relied upon for diagnosis. Given these constraints, the present study proposes diagnostic approaches compatible with this production system's realities.
Serological analysis determined the presence of antibodies against Leptospira spp. being significantly higher (p<0.05) in non-pregnant females compared to pregnant females. The serovars and titers found could be consistent with vaccinations or even with previous infections, making it impossible to determine the origin of these antibodies with the technique used (MAT) (Mummah et al. 2024). It should be noted that 80% of the seropositive cattle had antibodies against 2 serovars present in commercial vaccines. The remaining 20% of seropositive cattle presented antibody for the serovar Hardjo, and although cattle could be the reservoir of this serovar, it may also cause disease, and those titers found could be considered significant enough for acute disease (Wilson Welder et al. 2021). Although high serology may indicate acute infection, certain levels, such as those observed in this case, could not be representative to indicate infection, requiring a paired sampling, showing the limitation of this technique.
For N. caninum, a high seroprevalence was found in this study in coincidence with previous reports in both cattle (Moore et al. 2014) and buffaloes (Konrad et al. 2013). In this case, the seroprevalence in pregnant females was higher than in aborted females, but no statistically significant difference was found. Camillo et al. (2010) and Pulido-Medellín et al. (2016) indicated that the vertical transmission route is the most frequent; in the present work, this transmission route was not evaluated. However, 42.8% of the aborted females changed their serological status during pregnancy, which may indicate horizontal transmission. However, it could be possible that during the first sampling the titers were lower than the cut-off value used in this test (1/100) due to the antibody fluctuations during gestation. Additionally, further studies, such as rNcGRA7- and rNcSAG4-based ELISAs, should be performed to discard persistent infected animals, whose titters could be low (Caspe et al. 2012, Guido et al. 2016, Fereig et al. 2021). According to Guarino et al. (2000) older animals are more likely to have been exposed to oocysts at least once during their lifespan compared to younger animals.
In 6 aborted females, 12 bulls and 3 fetuses, the agents were identified as responsible for some of the observed losses by culture of CVM, PS and fetal analysis. In 6 aborted females, 12 bulls and 3 fetuses whose agents were identified by culture of CVM, PS and fetal analysis, these would have been responsible for some of the observed losses. Despite finding concomitant antibodies against other infectious agents and based on the aforementioned criteria, it was determined that these antibodies could possibly be due to the response of an immunocompetent fetus and not to the cause of death.
According to Truyers et al. (2014), abortions by C. fetus and T. foetus can occur at any time but are most frequently detected between 4 and 6 months of gestation. This could be the case of the remaining 55 aborted females, where the cause could not be established, but some of them shared herd with females with positive diagnosis. However, there is also a possibility that those cases not having reached the diagnosis in these 55 cases, losses, since they could be associated with other infectious or non-infectious causes, which, with mucus culture alone, cannot be established, were not included in the study. In this sense, some authors indicated that traditional diagnosis methods based on culture and serology can be relatively low sensitive, depending on several factors that can affect the diagnosis such as the quality of the sample and the period elapsed since the sampling and the arriving to the laboratory (Wheelhouse et al. 2012, Kalogianni et al. 2021, Garcia et al. 2022, Zhou et al. 2022). When the CVM of aborted cows was analyzed, venereal agents were found responsible for some losses. However, analyzing aborted fetuses, the existence of other diseases within the herds was confirmed, settling the importance of concomitant fetal analysis.
The finding in this study of females possibly infected by horizontal transmission by N. caninum opens the debate as to whether transmission in these farms could be greater than vertical transmission, and the need to evaluate the reason why seroprevalence is higher in north-western Argentina than in other areas of the country.
The present study confirms the importance of the analysis of pre-service bulls, as well as the study of aborted cows and fetuses, to detect the cause of loss.
In conclusion, regarding the extensive production systems of northeastern Argentina, where recovery of aborted fetuses is uncommon, serology and bacteriological sampling represent practical diagnostic approaches. The findings of this study contribute improve the limited current knowledge of reproductive losses in the Mesopotamian region and provide a basis for future studies, which may address a broad spectrum of agents such as viral pathogens and the potential horizontal transmission of Neospora caninum in the context of wildlife-cattle interactions.
Acknowledgments. This study was funded by Instituto Nacional de Tecnología Agropecuaria (INTA) grant: Structural Project 2023-PE-L01-I050.
Statement of animal rights. The experimental procedures were performed in compliance with the Comité Institucional para el Cuidado y Uso de Animales de Experimentación (CICUAE, INTA-, Argentina): Aval.
Authorship contribution statement. DRP: Investigation, methodology, formal analysis, writing – original draft, writing – review and editing. SJM: Investigation, funding acquisition, writing – original draft, writing – review and editing. MV: Data curation Prepared the samples, writing – original draft. BD: Investigation, writing – original draft, writing – review and editing. CSG: Formal analysis, writing – original draft, writing – review and editing
Declaration of competing interest. The authors declare that they have no conflict of interest.
Data availability. Data will be made available on request.