Revista Veterinaria. 2026; 37(1)
https://doi.org/10.30972/vet.3719100
Artículo Original




Recibido: 14 octubre 2025 / Aceptado: 22 diciembre 2025
Abstract
Pregnancy toxemia, also known as pregnancy-associated systemic disease (PASD), is a metabolic disorder affecting sheep and goats during the later stages of gestation, particularly in animals carrying multiple fetuses. This study describes seven outbreaks of pregnancy toxemia in small ruminants in northwestern and southern Argentina, diagnosed at the Animal Health Laboratories of INTA Salta and INTA Bariloche. Morbidity rate ranged from 3% to 24%, and mortality reached up to 100% among affected animals. Necropsy and histopathological examinations revealed severe hepatic lipidosis, with up to 80% of the liver parenchyma affected. Elevated hepatic enzymes and the presence of urinary ketone bodies confirmed the diagnosis in two outbreaks. Goats exhibited more severe lesions and a poorer therapeutic response than sheep. The disease occurred under both adequate and deficient nutritional conditions, suggesting the influence of multiple predisposing factors. These findings underscore the importance of appropriate early nutritional management and preventive monitoring during late gestation, and provide new regional data that contribute to a better understanding of pregnancy toxemia in small ruminants.
Key words: metabolic disease, small ruminants, hepatic lipidosis, negative energy balance.
Resumen. La toxemia de la preñez es un trastorno metabólico que afecta a ovejas y cabras durante las últimas etapas de la gestación, especialmente a aquellas que llevan múltiples fetos. Este artículo describe siete brotes de toxemia de la preñez en pequeños rumiantes del noroeste y sur de Argentina, diagnosticados en los Laboratorios de Salud Animal de INTA Salta y de INTA Bariloche. La tasa de morbilidad varió entre 3% y 24% y la mortalidad alcanzó hasta el 100% de los animales afectados. Los hallazgos de necropsia e histopatología revelaron lipidosis hepática severa con hasta un 80% de afectación del parénquima. La elevación de enzimas hepáticas y la presencia de cuerpos cetónicos en orina confirmaron el diagnóstico en algunos casos. Las cabras presentaron lesiones más graves y una respuesta terapéutica inferior que las ovejas. La enfermedad se presentó tanto en condiciones nutricionales adecuadas como deficientes, lo que sugiere la influencia de múltiples factores predisponentes. Estos resultados resaltan la importancia de un adecuado manejo nutricional temprano y un monitoreo preventivo durante la gestación avanzada y aportan nuevos datos regionales sobre la toxemia de la preñez en pequeños rumiantes.
Palabras clave: enfermedad metabólica, pequeños rumiantes, lipidosis hepática, balance energético negativo.
Pregnancy toxemia is a critical metabolic disorder affecting sheep and goats during late gestation. This condition is characterized by partial anorexia, depression and often neurological signs that progress to recumbency and death. The disease primarily affects animals carrying multiple fetuses and is typically seen in those with additional risk factors such as inadequate nutrition or other underlying health issues. The economic and welfare impacts of pregnancy toxemia on livestock production make it a significant concern for veterinarians and farmers alike, particularly in regions where small ruminants play a crucial role in agricultural systems (Mongini and Van Saun 2023).
The pathogenesis of pregnancy toxemia is closely linked to the metabolic demands of late gestation, where the growing fetuses significantly increase the mother's energy requirements. Inadequate nutrition during this critical period leads to a negative energy balance, prompting excessive mobilization of fat stores. This overwhelms the liver’s capacity to metabolize fat, resulting in hepatic lipidosis and a cascade of metabolic disturbances. The body condition score and the number of fetuses influence the severity and progression of the disease (Ji et al. 2023).
In Argentina, as in other countries, the prevalence of pregnancy toxemia varies depending on management practices, climatic conditions and the nutritional status of the animals. Despite the global occurrence, there are regional differences in the clinical presentation and outcomes, highlighting the importance of localized studies to better understand and manage this condition in specific contexts (Kelay and Assefa 2018). The objective of this paper was to present a series of clinical cases of pregnancy toxemia in sheep and goats in Argentina, providing insights into the regional expression of this complex disorder.
This study included seven outbreaks of pregnancy toxemia in sheep and goats from northwestern and southern Argentina, diagnosed through two laboratories belonging to the INTA National Veterinary Laboratory Diagnostic Network. The participating diagnostic centers were the Regional Service of Animal Health (SIRSA) at INTA Bariloche and the Animal Research Institute of the Semi-Arid Chaco (IIACS) at INTA Salta. For each outbreak, available clinical, epidemiological, and necropsy information was compiled and analyzed.
Because most animals were submitted dead to the laboratory, clinical data were obtained primarily from anamnesis provided by the attending veterinarians or producers, including information on treatments administered and observed responses. Access to live animals was possible only in a few cases (n = 4) from two outbreaks. In these animals, urine samples were collected to assess ketonuria using a semiquantitative urine dipstick test (Combur Test®, Wiener), following the manufacturer’s recommendations. Given the limited number of live animals and the advanced condition of many cases, biochemical testing could not be performed in all outbreaks.
All necropsied animals underwent complete post-mortem examination. Tissue samples from liver, kidney, brain, and other major organs were fixed in 10% neutral buffered formalin, routinely processed, and stained with hematoxylin and eosin (H&E) for histopathological evaluation. The presence and severity of hepatic lipidosis were assessed using standard histopathological criteria (Souto et al. 2019). In several outbreaks, carcasses arrived without prior clinical management, limiting the availability of treatment-related data.
This integrative approach allowed the combined evaluation of clinical, epidemiological, and pathological information despite the inherent limitations associated with post-mortem submissions and restricted access to live animals.
The morbidity ranged from 3 to 24%, whereas the lethality varied between 0 and 100% (Table 1). Most outbreaks occurred in late gestation, and all necropsied females carried one or more fetuses. Consistent gross findings included marked fatty liver and, in some cases, evidence of fat mobilization in omental or perirenal fat deposits (Figure 1).
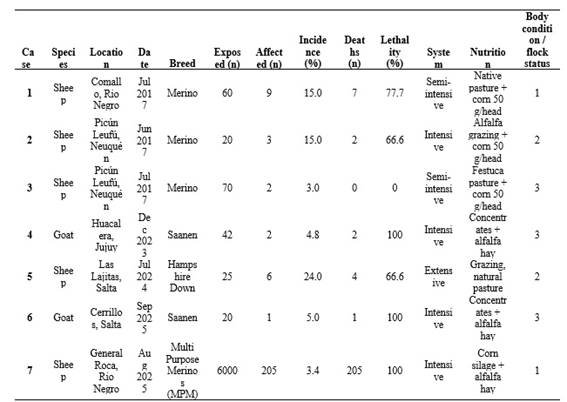
Histopathological examination confirmed the presence of both macrovesicular and microvesicular hepatic steatosis, with extensive parenchymal involvement reaching up to 80% of the hepatic tissue in some animals (Figure 1; Table 2).
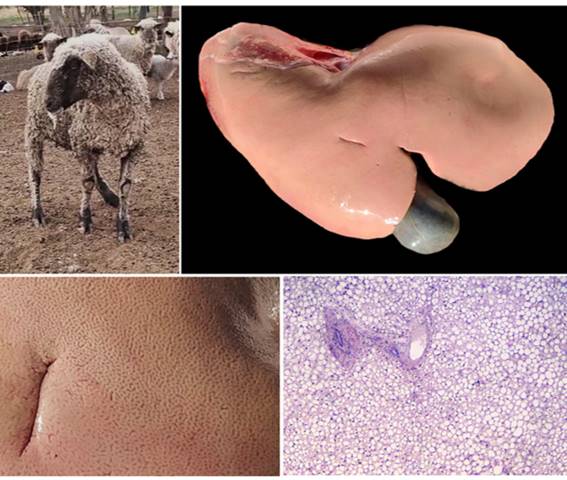
Macrovesicular steatosis was characterized by one or a few large, optically empty intracytoplasmic vacuoles that displaced the hepatocyte nucleus toward the cell periphery. In contrast, microvesicular steatosis consisted of numerous, small, uniform cytoplasmic vacuoles that did not displace the nucleus, which remained centrally located. Frequently, both patterns coexisted within the same lobular region, affecting periportal, midzonal, and centrilobular hepatocytes to varying degrees. The distribution of both lesion types was multifocal to coalescing, and in severely affected areas, hepatocytes were markedly swollen due to the accumulation of lipid vacuoles. In four of the outbreaks, ketone bodies were detected in urine, further supporting the diagnosis of pregnancy toxemia. Only in one outbreak were elevated hepatic enzymes detected in blood (Table 2).
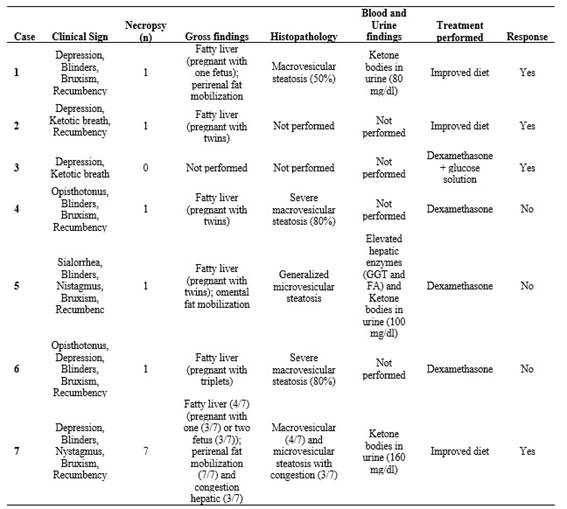
This study confirms the association between a negative energy balance in the latter stages of pregnancy and the presence of multiple fetuses in the cases of pregnancy toxemia (Brozos et al. 2011). Most of the affected animals were pregnant with twins or triplets, whose energy demands likely exceeded the available dietary supply. Interestingly, the syndrome was observed in flocks with good nutritional status that were correctly fed with concentrates and alfalfa hay, as well as in flocks with poor body condition and limited energy supplementation (Table 1). This variability suggests that many factors such as undernutrition, nutritional imbalances, abrupt dietary changes or excessive fat reserves may predispose animals to excessive hepatic lipid accumulation (Rook 2000, Bani Ismail et al. 2008). Similar situations have been documented in sheep flocks subjected to periods of nutritional restriction or adverse climatic conditions, where group-level stressors have been associated with increased morbidity and decreased overall performance. However, descriptions of the syndrome in goats are less frequent and isolated reports often highlight the severity of clinical progression (de Melo et al. 2009, Vasava et al. 2016). The clinical course in this species appears to be more acute and less responsive to therapy (Bani Ismail et al. 2008).
Pregnancy toxemia is characterized by profound metabolic disturbances, most notably a severe negative energy balance that leads to pronounced ketonemia. This condition is considered one of the disease's key biochemical hallmarks (Brozos et al. 2011, Ji et al. 2023). Affected animals typically exhibit elevated β-hydroxybutyrate concentrations, hypoglycemia, and varying degrees of hepatic dysfunction associated with extensive lipid mobilization. Consistent histopathological studies demonstrate diffuse hepatic lipidosis, reflecting these metabolic derangements (Souto et al. 2019). While such biochemical evaluations are essential for diagnosis, they could only be performed in a limited number of cases in the present study. Many animals were deceased at the time of examination, and postmortem changes restricted the applicability and reliability of laboratory testing. From a pathological standpoint, the gross and histopathological findings in this study-characterized by severe hepatic lipidosis with macro- or microvesicular steatosis-are consistent with traditional disease descriptions (Smith and Sherman 2022). However, the predominance of severe macrovesicular patterns in goats contrasts with ovine cases reported in the literature, which more commonly describe diffuse microvesicular infiltration (Mongini et al. 2023). The detection of ketone bodies in the urine of some animals and elevated liver enzymes (Table 2) aligns with previously published biochemical markers of the disease (Brozos et al. 2011). While clinical signs were recorded in all affected animals, they were not collected following a standardized or systematic protocol. Nevertheless, the combination of depression, recumbency, neurological signs, and poor response to treatment is consistent with the clinical presentation typically reported in ovine pregnancy toxemia (Mongini et al. 2023).
Treatment outcomes in the present series were variable, with four of the seven affected animals showing a favorable response. It is important to note that all cases occurred under field conditions, without hospital-based supportive care, and treatments were administered either by the attending farm veterinarians or directly by the producers. These circumstances inherently limit the intensity and timeliness of therapeutic interventions, which likely contributed to the overall poor outcomes observed. In this context, the treatment protocols employed in the evaluated flocks should indeed be reconsidered, as their application under field constraints may explain the high lethality and limited clinical effectiveness. The main therapeutic interventions were dexamethasone, fluid therapy, and dietary adjustments. However, recovery was only achieved in flocks where energy supplementation was corrected quickly. This finding aligns with the well-documented limited efficacy of pharmacological treatments in cases with advanced clinical signs (Navarre and Pugh 2009). As in previous reports, administering glucose or propylene glycol temporarily improved the condition, but it rarely altered the outcome if given too late (Marteniuk and Herdt 1988). The goats in this study appeared particularly unresponsive, a finding supported by Rook (2000), who noted interspecies differences in lipid metabolism and disease progression. Other authors have emphasized that intensive management systems with high-concentrate diets can paradoxically lead to metabolic disturbances in goats (Al-Qudah 2011), which may help explain the observed outcomes.
Pregnancy toxemia is a well-known disease worldwide, particularly in countries where goat and sheep production is relevant (Mongini et al. 2023). However, bibliographic records in Argentina are limited (Robles 2018). There are isolated reports from Patagonia associated with inadequate nutrition and stressful weather conditions and high mortality rates in sheep (Martínez et al. 2018). The disease has been described more frequently in neighboring countries. In Uruguay, for example, numerous cases have been reported in extensive production systems, particularly in ewes carrying multiple fetuses and during cold winters (Cáceres 2024). In Brazil, the disease is recognized as one of the main metabolic causes of mortality in small ruminants. Experimental and field studies have detailed its pathogenesis and management (Cavalcanti Souto and Bastos Afonso 2023, da Silva Júnior et al. 2025). In Chile, outbreaks have been reported in southern sheep flocks, where inadequate energy supplementation during the last third of gestation is a determining factor (Duarte and Williams 2003). These regional antecedents underscore the significance of addressing pregnancy toxemia as a health issue with substantial economic and productive implications in Southern South America, despite the limited formal documentation in Argentina.
From a preventive perspective, pregnancy toxemia is one of the most challenging metabolic disorders in small ruminants due to its multifactorial nature. A consistent risk factor across all outbreaks was late gestation in females with multiple fetuses. During this period, nutritional requirements increase dramatically (Ji et al. 2023). Therefore, preventive strategies should focus on carefully monitoring body condition, avoiding abrupt dietary changes and ensuring adequate, balanced energy intake during the last six weeks of gestation. Ultrasonography for fetal counting and subsequent dietary adjustment has been recommended to minimize risk (Souto et al. 2019, Mongini et al. 2023). This article emphasizes the importance of flock-level management, early veterinary intervention, and proactive nutritional planning to reduce the incidence and severity of pregnancy toxemia in sheeps and goats.
CRediT authorship contribution statement. SVG.: Investigation, Formal analysis, Data curation. MA.: Conceptualization, Resources, Supervision, Funding acquisition, Writing - review & editing. M. Felice: Investigation, Methodology, Data curation. L. Homse: Investigation, Validation, Resources. CCLA.: Investigation, Formal analysis, Visualization. MVDMN: Investigation, Methodology, Data curation. ACA.: Investigation, Formal analysis, Data curation. ALS: Investigation, Validation, Data curation. MJF: Conceptualization, Writing - original draft, Writing - review & editing, Supervision, Funding acquisition.
Declaration of competing interest. The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability. Data will be made available on request.