Revista Veterinaria. 2026; 37(1)
https://doi.org/10.30972/vet.3719286
Artículo Original




c4
 melinasimoncini22@yahoo.com.ar
melinasimoncini22@yahoo.com.ar
Recibido: 26 noviembre 2025
Aceptado: 03 abril 2026
Abstract
Precise temperature control during artificial incubation is a critical factor for optimizing embryonic development and minimizing morphological deformities in aquaculture. This study aimed to design, develop, and technically evaluate two artificial incubation systems capable of managing distinct thermal profiles, followed by an in vivo biological validation. The first prototype used thermoelectric Peltier technology to create a portable microenvironment. Although promising, it required high energy consumption to achieve a minimum temperature of 7.3 °C in a 5-liter volume, indicating operational limitations for strict biological standards at larger scales. Consequently, a second device was developed: a closed-loop, double-layer incubator integrated with an IoT-based telemetry system (ESP8266 microcontroller and Telegram bot). This system successfully maintained highly stable thermal environments across four experimental setpoints (6.2 °C, 11.6 °C, 17.6 °C, and 23.5 °C) with temperature fluctuations below 1 °C. To validate the practical utility of this system, an in vivo proof-of-concept trial was conducted using Carassius auratus eggs under seven thermal treatments. The biological results confirmed that the system's thermal precision translates into reliable experimental data, demonstrating highly significant differences in hatching time, hatching rate, and observable morphological abnormalities (p<0.001), particularly under cold stress conditions. In conclusion, although the Peltier-based prototype requires further optimization, the double-layer incubator proved to be a highly accurate, scalable, and versatile tool that effectively reduces stochastic thermal variability in applied aquaculture research.
Key words: Carassius auratus, closed-loop system, microcontroller, incubation systems, Peltier, remote monitoring.
Resumen. El control preciso de la temperatura durante la incubación artificial es un factor crítico para optimizar el desarrollo embrionario y minimizar las deformidades morfológicas en la acuicultura. Este estudio tuvo como objetivo diseñar, desarrollar y evaluar técnicamente dos sistemas de incubación artificial capaces de gestionar distintos perfiles térmicos, seguido de una validación biológica in vivo. El primer prototipo utilizó tecnología termoeléctrica Peltier para crear un microambiente portátil. Si bien era prometedor, alcanzar una temperatura mínima de 7,3 °C en un volumen de 5 litros requería un alto consumo de energía, lo que indica límites operativos para estándares biológicos estrictos a escala. En consecuencia, se desarrolló un segundo dispositivo: una incubadora de circuito cerrado de doble capa integrada con un sistema de telemetría basado en IoT (microcontrolador ESP8266 y bot de Telegram). Este sistema mantuvo con éxito entornos térmicos altamente estables en cuatro puntos de ajuste experimentales (6,2 °C, 11,6 °C, 17,6 °C y 23,5 °C) con fluctuaciones continuas inferiores a 1 °C. Para validar la utilidad práctica de este sistema de doble capa, se realizó una prueba de concepto in vivo complementaria con huevos de Carassius auratus en siete tratamientos térmicos. Los resultados biológicos confirmaron que la precisión térmica del equipo se traduce directamente en datos fiables, demostrando diferencias altamente significativas en los tiempos de eclosión, las tasas de eclosión y las malformaciones físicas evidentes (p<0,001), especialmente en condiciones de estrés por frío. En conclusión, si bien el prototipo Peltier requiere mayor optimización, la incubadora de doble capa demostró ser una herramienta altamente precisa, escalable y versátil que elimina eficazmente las variaciones térmicas aleatorias para la investigación aplicada en acuicultura.
Palabras clave: Carassius auratus, sistema de circuito cerrado, microcontrolador, prototipos de incubadoras, Peltier, monitoreo remoto.
Aquaculture consists of the construction of a controlled artificial aquatic environment, with the purpose of maintaining or reproducing ichthyic species or species in aquatic habitats. The Food and Agriculture Organization of the United Nations (FAO) defines aquaculture as an activity aimed at producing and fattening aquatic organisms in their environment. Global aquaculture production has grown steadily in recent decades. The increase was 6% per year in the 1960s, while growth reached 50% in the 2010s. In 2018 specifically, it reached a historical record of 114.5 million tons live weight, with a total farm sales value of USD 263.6 billion, and global per-capita consumption exceeding 20 kg (FAO 2020). Aquaculture production has become a primary source of economic value, recently surpassing capture fisheries in total production (FAO 2024). Within this sector, the ornamental fish industry represents a significant and rapidly growing global market, demanding high levels of technical precision to ensure quality and survival. Available data on the situation in Argentina show sustained growth in sectoral exports over the last 20 years (Carciofi et al. 2022).
The constant incorporation of technology in aquaculture has made it possible to implement new ways to contribute to the sustained development of this sector (Nederlof et al. 2021, Sánchez-Calle et al. 2021, Cargnin et al. 2022). However, the increase in aquaculture production has not been homogeneous across species, as some are easier to farm than others. Also, the aquaculture sector is able to adapt more quickly and efficiently to changes in consumer preferences, as aquaculturists increasingly exercise more control over their production (FAO 2020). Existing knowledge of both the biology and ecology of many aquatic organisms allows for a rapid adaptation of technology and techniques that improve production. The use of innovative tools to understand larval developmental biology is allowing for the precise manipulation of growth parameters (Motta et al. 2023, Honji et al. 2025). Innovations, undoubtedly, allow for the optimization of resources and production processes (Nederlof et al. 2021, Sánchez-Calle et al. 2021, Cargnin et al. 2022).
The control of water physicochemical parameters and classification algorithms in fish have achieved time optimization, reduced costs and reduced systematic errors (Sánchez-Calle et al. 2021). In particular, temperature is one of the parameters over which greater control can be exercised, which has a direct and significant impact on production. It is considered one of the two variables, together with photoperiod, that most affect the development of aquatic organisms due to its importance in the control of physiological processes (Fuiman and Werner 2009). The length of the embryonic period and the rate of yolk consumption are strongly influenced by the temperature of the water surrounding the spawn (Ramos et al. 2025). For example, in Carassius auratus (Linnaeus 1758) the rate of cell cleavage accelerates rapidly as temperature increases. Synchronized cell divisions occur at 131-min intervals at 10 °C, at 33-min intervals at 20 °C, and at 19-min intervals at 30 °C during the scission period (Urushibata et al. 2020). The rate of abnormal embryos hatched increase significantly at high temperatures, with development being unviable above 34 °C, whereas no differences in the number of abnormal embryos are detected in groups of eggs incubated at temperatures near 24 °C (Urushibata et al. 2020, Motta et al. 2023). At this temperature, eggs hatch in 76 hours (Motta et al. 2023). Conversely, exposure to suboptimal low temperatures, or cold stress, drastically disrupts embryonic development, leading to severe hatching failures and irreversible morphological abnormalities (Zhang et al. 2023). To achieve this precise temperature management, various engineering approaches can be implemented in artificial incubators. Thermoelectric cooling, or Peltier technology, has emerged as a compact and portable alternative capable of creating controlled micro-environments (Cheepati and Balal 2024). It operates by transferring heat from one side of the device to the other using electrical energy, although its thermal efficiency in larger volumes requires careful evaluation (Enescu and Virjoghe 2014). Alternatively, double-layer incubation systems - which use a closed-loop environment within an isolated macro-chamber - offer significant thermal stability by buffering external temperature fluctuations (Tan and Zhang 2024). This dual-barrier approach is highly effective for maintaining the strict temperature tolerances required by sensitive biological materials. Despite their potential, the specific application of these systems to manage the hatching times of ornamental fish remains underexplored. Therefore, our objective was twofold: first, to design and develop two device options for precise control of different incubation temperatures; and second, to biologically validate these systems by evaluating the effects of varying thermal treatments on egg hatching times, hatching rates, and the incidence of physical malformations.
Two experiments were conducted to program devices to control four experimental temperatures, three of which (6 °C, 12 °C and 18 °C) controlled a moderate volume and the fourth temperature (24 °C) controlled a large volume. Three yield evaluations were performed, with an experimentation time defined as 14 days for each yield evaluation.
Device 1
Peltier. In the first instance, a portable prototype with Peltier technology was experimented with. This device generates a thermoelectric effect by converting the temperature difference to electrical voltage and vice versa; it comprises three separately identified effects, the Seebeck effect, the Peltier effect, and the Thomson effect. Specifically, the Peltier effect performs the inverse action of the Seebeck effect. It consists in the creation of a thermal differential from an electrical potential difference. This occurs when a current passes through two dissimilar metals or semiconductors that are connected in two solder joints (Peltier junctions). The current produces a heat transfer from one junction, which cools down, to the other, which heats up (Mardini-Boveaet al. 2019).
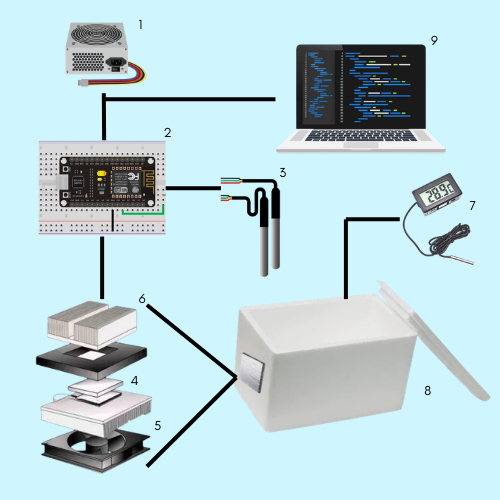
The experimental prototype (Figure 1) was composed of a 12,708 Peltier cell, a cooling kit (90 x 140 x 40 mm and 60 x 45 x 20 mm metal heat sinks of 12 V 120 W, together with an 80 x 80 x 15 mm cooler and an adhesive insulating rubber) regulated by a Mosfet P-55v-74a transistor, either a 12 V 9 A or a 12 V 20 A switching source that supplied electrical power (sources used separately), a submersible digital temperature sensor connected to a microcontroller (which recorded and stored data during the period in which the previously programmed commands were executed), a digital thermometer, and two Styrofoam containers. The first container had a thickness of 1 cm, was 12 cm in diameter and 15 cm high with an internal volume of 1 liter and did not contain water bottles. The second container had a thickness of 1 cm and dimensions of 24 x 24 x 40 cm, featured an internal thermal insulation of aluminized polyurethane (1 cm thick), and contained 5 liters of water along with two glass jars (3 mm thick, dimensions 7.5 x 14 x 7.5 cm) holding 0.5 liters of water each
Initially, experiment A was carried out with a 1-liter container without water content. Eighteen attempts were replicated, each with a duration of one hour and the temperature was recorded every 15 minutes. Finally, experiment B was conducted with a 5-liter container and two glass jars (3 mm thick and dimensions 7.5 x 14 x 7.5 cm) with 0.5 liters of water, and a 12 V and 20 A source (to provide more power to the equipment). Eighteen replicates were carried out, two replicates of two hours each per day, with a temperature recording every 15 minutes. After each replicate in experiment "A" and "B," the temperature was recorded after 30 minutes without the Peltier operating in order to identify the heat gained by the vessel (Figure 1).
The operation of the equipment for both tests is designed as a closed loop system, which takes the process output and compares it with the reference signal in order to monitor the evolution of the variable at all times. For this first unit, the switching source supplies electric power, and a microcontroller integrates the electro-regulatory function of the Mosfet through the processing of information captured by the digital temperature sensor. In this way, the parallel semiconductors of the Peltier cell are stimulated and generate heat on the external side while the internal side perceives the absence of heat.
The microcontroller has Firmware of our authorship, developed in C++ (Trott 2019) using object-oriented programming. Atom (Szabo 2024) was used as an integrated development environment, and PlatformIO (Loginov 2018) as a platform for embedded development. For the notifications and alerts system, Telegram was used with its own bot created for this purpose. The connection was made through calls to Telegram's HTTPS REST API. For the telemetry system, Google Sheets was used with a self-developed Google script in proprietary language (from Google) based on JavaScript, capable of receiving HTTPS calls from the microcontroller and translating them into the creation of a new data entry. All the software used is open source or otherwise freely available.
The Peltier equipment was mounted in a 60 x 45 mm slot on the narrow right side of the Styrofoam container, where only the metal heat sink that makes contact with the cold side of the Peltier cell enters. The edges of the slot were sealed with high-temperature FASTIX®. The heat sink was isolated from the incubator, with the cooler mounted on the metal filaments to facilitate heat radiation.
The operation of the equipment started with the microcontroller that recorded the water temperatures through the digital temperature sensors, and based on the programmed temperature, allowed the Mosfet to give continuity to the electric current coming from the switching source to feed the Peltier cell. The cell operated until the desired temperature was reached, which continued to be monitored and regulated by the sensors and the microcontroller. When the programmed temperature was reached, the microcontroller modified the state of the Mosfet to prevent the electric current from continuing to flow to the Peltier. In this way, every certain period, there were complete cooling cycles and a subsequent "rest" of the Peltier, thus keeping the internal temperature constant.
Device 2
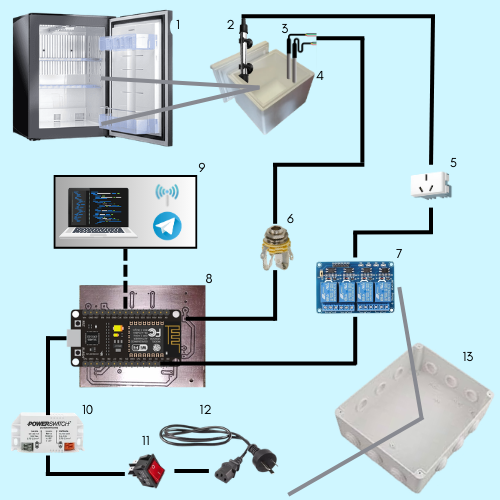
Double-layer incubator. First layer delimited by a Briket® Undercounter Refrigerator (146 l, 1010 Lared). In this way, the containers inside the equipment were subjected to homogeneous conditions of temperature and humidity. The equipment was conditioned, removing part of its internal structure and leaving only what was necessary to mount the incubation receptacles. The refrigerator temperature for this first layer was set at 2-3 °C and remained at this temperature throughout the experiment, as the minimum incubation temperature to be tested in a second layer was 6 °C. Temperature measurements were taken with 4 digital sensors located inside the refrigerator and the door was opened for 5 minutes in a room at 24 °C (temperature recorded by a fifth sensor not integrated into the microcontroller). The procedure was performed 4 more times and in each repetition the sensors were rotated. The data acquisition was through a Nodemcu (Esp8266) which was connected to the 4 sensors. The subsequent analysis to obtain the average was by way of a spreadsheet (Figure 2).
The second layer is defined by four incubation containers (1-cm-thick styrofoam containers with lids), three of which were of equal dimensions, 12 x 12 x 34 cm (total volume of 5 l) and a fourth of 24 x 24 x 40 cm (total volume of 23 l). Inside each container, glass jars without lids (where the eggs to be incubated would eventually be placed) were placed, one for each experimental level.
The incubator was planned so that the refrigerator cools all the receptacles. The receptacles have a 3 cm2 slot on the narrow right side, through which the water heater cables enter and exchange temperatures, as well as the siliconized tube of the aerator and the digital temperature sensor. The containers have a volume of water that occupies half of their capacity. Internally, the glass jars, a water heater (10 W mini aquarium heater), a digital temperature sensor and the respective tubes that conduct the oxygen to each glass jar and the surrounding water were incorporated.
The function of the aerator in the jars is to provide oxygen to the eggs and in the surrounding water it generates turbulence and homogenizes the water temperature. At the point where the sensors register a lower temperature than desired, the heaters (activated by the microcontroller in the grey box) compensate in a controlled manner by supplying heat.
The sensors were tested and found to have a precision error of +/- 0.2-0.3 °C. They are located with elastic bands on the outer narrow side of the glass container in front of the aquarium heater. All their data is processed by a microcontroller inside the grey box and stored in the cloud (account and personal drive folder; extension ‘.xls’).
The grey box or smart shoe was designed to integrate the complete control of the system in its second layer. It consists of a watertight box suitable for electrical connection (24 x 30 x 8 cm), interlock plug connector for chassis, 10 A single-pole switch with neon, 12 V 1 A switching power supply, 4 optocoupled relay module, printed circuit board (PCB) of our own design and manufacture (consisting of a NODEMCU development board based on the ESP8266 microcontroller family, 5 V voltage regulator model 7805 and 2.54 mm micro pin connectors with cable), 4 TRS 6.5 mm jack connectors for chassis, 4 temperature sensors (consisting of 4 submersible temperature sensors model Ds18b20, 4 4700-Ohm resistors and 4 Plug TRS 6.5 mm cable connectors) and finally 4 plug-in 220 V 10 A sockets (Figure 2 ).
The internal functioning of the grey box is homologous to that of the Peltier system, where the switching source electrically powers the entire circuit and the logic unit is the microcontroller. The microcontroller receives and processes the temperatures registered by the four temperature sensors (located in their respective incubation receptacles), and also relays the data to the cloud (Google Sheets). When temperatures lower than those preset in the microcontroller are detected, the relay channel(s) that allow the continuity of the electrical current to each of the 4 outlets to which the aquarium heaters are plugged, are activated. These devices raise the temperature of the surrounding water inside the incubation vessels until the ideal incubation temperature is reached in each one. At this point, again via the temperature sensor, the microcontroller intervenes on the channel in question to interrupt the flow of current to the outlets and thus to the water heaters. This generates constant cycles of heating and cooling of the body of water, dynamically and autonomously maintaining the temperature constant in each incubation receptacle.
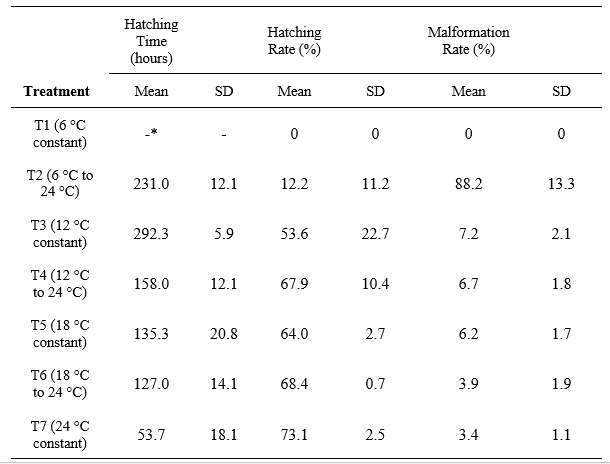
Preliminary Biological Validation. To validate the practical functionality of the double-layer incubator, an in vivo proof of concept was conducted using Carassius auratus eggs. The experiment used the four baseline working temperatures of the device (6 °C, 12 °C, 18 °C, and 24 °C) to establish seven distinct thermal treatments. Four of these treatments maintained a constant temperature throughout the entire incubation period until hatching (6 °C, 12 °C, 18 °C, and 24 °C). The remaining three treatments were subjected to a thermal shift to evaluate varied profiles: eggs were initially incubated at 6 °C, 12 °C, and 18 °C for five days, and subsequently transferred to the 24 °C incubator until hatching. Three replicates of 75 fertilized eggs per treatment were placed in the device's incubation jars. The primary goal was to confirm the equipment's capacity to sustain biological trials by evaluating three key response variables: hatching time, hatching rate, and the percentage of evident physical malformations in the resulting larvae.
Statistical analysis. All statistical analyses were performed using RStudio software version 4.1.1 (R Core Team 2021), operating within the RStudio integrated development environment. Data visualizations were generated using customized R graphics libraries. Additionally, boxplots for thermal distribution were created using the Python programming language, utilizing the Pandas, Matplotlib, and Seaborn libraries. The performance of the incubation devices (temperature stability and fluctuations) was evaluated strictly through descriptive statistics (means and standard deviations). Conversely, to evaluate significant differences in the biological validation - specifically regarding hatching time, hatching rate, and the percentage of evident physical malformations across thermal treatments - one-way analysis of variance (ANOVA) models was utilized. When significant differences were detected, a Bonferroni post-hoc test was applied using a significance level of p<0.05.
Experimentation with Peltier cells for the development of portable incubators. During the initial evaluation of the Peltier technology (Experiment A, using a 1-liter empty container and a 12 V 9 A power source), the equipment failed to reach the target temperatures. The average initial temperature was 22.7 °C, and the absolute minimum temperature recorded during the trials was 13.4 °C. After averaging the values obtained every 15 minutes across all replicates, the mean minimum temperature reached after one hour of operation was 14.65 °C (Figure 3).
Furthermore, with a recorded energy consumption of 55 W, the cooling rate of the Peltier cell decreased significantly as it approached one hour of continuous operation. The average temperature reduction (ΔT) during the first 15 minutes was -2.68 °C, but this rate dropped to -0.45 °C in the final 15 minutes, meaning the thermal efficiency was reduced to approximately one-sixth of its initial performance. Additionally, to evaluate the thermal insulation, temperature was recorded 30 minutes after turning off the Peltier device; the vessel exhibited an average heat gain of 4.6 °C.
Given these initial limitations, system components were modified for Experiment B. The thermal insulation on both sides of the Peltier cell was improved, a higher amperage power source (20 A) was implemented, and the system was tested in the larger 5-liter container containing water-filled jars. Under these modified conditions, the system achieved values closer to the desired targets. The average initial temperature was 18.2 °C, and the absolute minimum recorded was 4.7 °C. Averaging the replicates every 15 minutes yielded a mean minimum temperature of 7.3 °C after one hour of operation (Figure 4).
Despite the improved thermal drop, the high energy cost and the operational limits of the Peltier technology at this scale indicated that reaching and steadily maintaining the strict thermal standards required for biological incubation would demand a disproportionate investment of resources, time, and budget. For this reason, the development focus was shifted entirely to the double-layer incubator system.
Experimentation with the double-layer closed loop system. The average values for each incubator inside the refrigerator were 6.2 °C for Incu01 (incubator one defined at 6 °C); 11.7 °C for Incu02 (incubator two defined at 12 °C); 17.7 °C for Incu03 (incubator 3 defined at 18 °C) and 23.5 °C for Incu04 (incubator four defined at 24 °C).
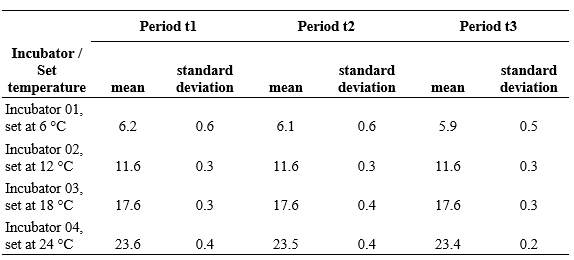
Once in operation, the records for each incubator showed a correct performance (Table 1), which met the expectations of maintaining constant temperatures with oscillations of less than one degree as long as the refrigerator is kept closed. The extreme values or isolated points (Figure 5) are values taken during a manual control period, while the refrigerator door was open and the ambient temperature ranged from 26 to 31 °C.
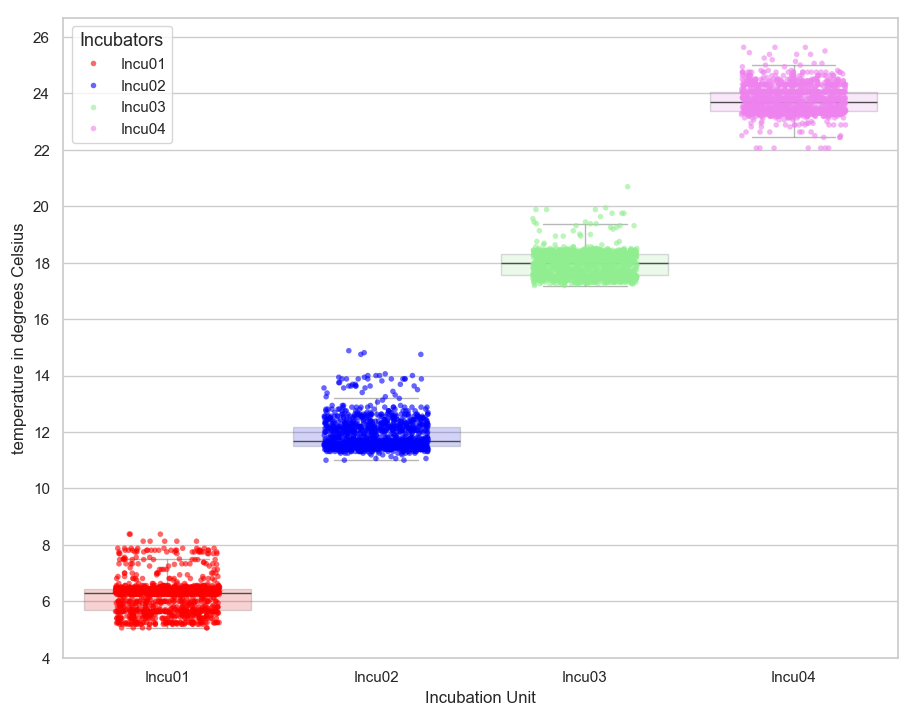
In the extremely hot months, the refrigerator worked at full power. With the help of a digital refrigerator thermometer, it was found that the unit, in forced operation, reached a minimum internal temperature of 8 to 9 °C during the midday and afternoon hours. This jeopardized the correct operation of the incubator at 6 °C. In response to this, the unit was separated from the wall and an air flow was supplied to the rear of the unit by means of a floor fan. Performance improved significantly, achieving minimum values of 3 to 4 °C during the midday hours.
Biological validation of the double-layer system. One-way ANOVA models confirmed that the incubation temperatures provided by the unit generated highly significant differences in all the parameters evaluated: hatching time, hatching rate, and evident physical malformations (p<0.001). Post-hoc Bonferroni tests revealed that the optimal temperature of 24 °C (T7) significantly reduced both hatching times and malformations compared to the other treatments. Furthermore, the strict thermal control successfully induced expected thermal stress responses; the Bonferroni test confirmed that the malformation rate in the T2 treatment (6 °C to 24 °C shift) was significantly higher than in all other viable treatments (p<0.05).
We consider that precise control of thermal and oxygenation conditions during artificial incubation not only regulates embryonic development but also minimizes morphological deformities compared to natural spawning, where variables are stochastic (Li et al. 2024, Wang et al. 2025). In particular, the control of incubation temperature is fundamental for the development of aquatic organisms (Fuiman and Werner 2009). Guided by these principles, we propose the functionalities of the developed prototypes.
Peltier Incubator Development
Considering the results, it can be inferred that both the power supply and the insulation between both sides of the Peltier cell have a substantial influence on its performance. In agreement with García-Sobrino (2020), who noted that the electrical power consumed by the Peltier cell has a quadratic dependence on current intensity, it is necessary to dissipate as much heat as possible from the external face to improve performance. Care must be taken to ensure that this heat dissipation does not compromise the cooling of the internal face. Otherwise, the supplied voltage must be increased to achieve the desired cooling, generating disproportionate and inefficient energy consumption.
García-Sobrino (2020) also concludes that in a controlled environment, the maximum temperature difference between the internal and external faces is 25 °C. This reinforces the need for more efficient heat dissipation on the external side to achieve cooling values below 13 °C on the internal side. Under these conditions, the Peltier cell is subjected to a workload that does not compromise its lifespan, given that the semiconductors are joined to a conductive material (such as copper) using a low-melting-point solder. One of the most used solder materials is an alloy of 58% Bi and 42% Sn, which melts at 138 °C. Consequently, the temperature of the hot face must remain below 100 °C to avoid structural damage to the Peltier cell (Villasevil-Marco 2006).
Furthermore, the attachment system to the vessel wall must be precise, and the free space between the two metal heat sinks must be properly insulated. This prevents unwanted thermal bridging between the heat sinks. By doing this, the small heat sink is contained within the vessel walls and internally insulated, remaining in contact solely with the cold side of the Peltier cell. These adjustments yielded better performance, decreasing the temperature of the small heat sink to a minimum of 4.7 °C, whereas prior to these modifications, the system only achieved a minimum of 13.4 °C. Although other modifications also contributed to these results, completely isolating the heat sinks is crucial to maximizing their performance.
Polyurethane bonded to other materials or sealed with paint reduces the rate of heat transfer by conduction through the walls, which directly impacts the energy consumption of the equipment and prevents internal temperature increases (Godoy-Vaca 2016). However, even with the appropriate modifications, energy consumption remains inefficient. Considering continuous consumption of 55 W per unit, operating four independent Peltier incubators simultaneously for the 14-day experimental period would result in a total energy consumption of approximately 74 to 80 kWh.
In contrast to these energy limitations, the equipment offers several advantages. First, it has no moving parts, requiring little to no maintenance, and it allows for temperature control within fractions of a degree. Additionally, its compact size offers excellent portability compared to standard refrigeration incubators. It can even operate on car batteries, facilitating its transport to rural areas or field sampling sites that require controlled refrigeration. In short, being lighter and smaller than ice-filled containers, it provides highly accurate, reversible temperature control adjustable by altering the direction and intensity of the electric current. Furthermore, Peltier cells are free of chlorofluorocarbons (CFCs) and boast a long lifespan, achieving a mean time between failures (MTBF) of over 200,000 hours under stable operating conditions (Ferrotec 2025). Finally, despite its operational inefficiency, its construction cost is significantly lower than that of commercial biomaterial incubators or cold-heat fermentation chambers.
For future work, we consider it necessary to upgrade the incubation vessel and the power supply, potentially incorporating two Peltier cells operating on the same vessel with minor air or water recirculation (using a cooler or compressor-type aerator). Controlling the ambient temperature to avoid external heat exceeding 30-35 °C would also be beneficial. In light of our results, designing and assembling a highly functional Peltier device is feasible, albeit requiring a greater investment of time and resources.
Development of a double-layer incubator. The development of the Telegram bot was a great success; it significantly facilitated the daily recording of temperatures without the need to open the refrigerator. The system successfully managed everything from executing commands via the mobile application to sending notifications for manual resets or power outages. This aligns with Parra-Hernández and Suarez-Sierra (2016), who stated that implementing monitoring technology during incubation considerably decreases the time, attention, and effort required to care for the biological samples.
The microcontroller operated correctly, successfully recording sensor data, storing information in the cloud, and modulating the electric current. This reliable performance was expected based on Intriago-Loor (2019), who reported that the signal transmission error with the ESP8266 is generally 3.3%, dropping to 1.7% in environments with low electromagnetic noise. This latter finding is significant, as it confirmed that the refrigerator's structure did not act as a Faraday cage, thereby allowing the microcontroller to communicate with the local network and link to the cloud. Its flawless operation at temperatures below 4 °C is also noteworthy.
After an exhaustive market search, we found no existing technological developments that meet all these specific needs simultaneously in a single piece of equipment. The prototypes closest to our design were those proposed by Parra-Hernández and Suarez-Sierra (2016) and Nájera et al. (2020). Future work should explore implementing a monitoring system to track microcontroller disconnections, gathering accurate data on power outages or specific hardware malfunctions. Similarly, gaining remote control and operational data from the individual water heaters would make it possible to optimize energy consumption and immediately detect malfunction. Furthermore, future multidisciplinary studies should focus on advanced thermodynamic modeling, including the calculation of specific heat transfer coefficients and performance under varying thermal loads, to fully characterize and optimize the system's physical capabilities.
Biological Validation and System Versatility. The biological trial using Carassius auratus confirmed the high reliability of the double-layer incubator for sustaining in vivo experimental research. The thermal precision of the device effectively modulated embryonic development, with descriptive statistics summarizing the biological responses across the seven treatments. This preliminary biological validation conducted as a crucial proof of concept demonstrated that the significant differences observed in hatching times, hatching rates, and physical malformations across the treatments directly translate into reliable and reproducible biological data.
This aligns perfectly with the recent findings of Zhang et al. (2023), who demonstrated that exposing fish embryos to suboptimal low temperatures-or cold stress-significantly disrupts embryonic development, leading to severe hatching failures, increased cellular oxidative stress, and irreversible morphological and neurological abnormalities. Specifically, our device demonstrated its capacity to strictly maintain optimal constant temperatures to maximize hatching success, while also executing controlled thermal shifts to accurately induce and study thermal stress responses. As Franke et al. (2024) highlighted, the early life stages of fish are extraordinarily sensitive to environmental fluctuations; therefore, strict adherence to specific temperature regimes is essential to accurately evaluate thermal stress responses without the interference of uncontrolled variables. This in vivo biological validation confirms that the hardware successfully eliminates the stochastic environmental parameters typically associated with poorly controlled artificial setups, offering a scalable and highly accurate tool for applied aquaculture research.
Furthermore, while the primary focus of this study was optimizing conditions for C. auratus, whose eggs are adhesive, the versatility of the designed prototype extends to a wide variety of aquaculture species. The broad thermal range successfully maintained by the double-layer system (from 6 °C to 24 °C) demonstrates its capacity to incubate cold-water species, such as trout, as well as warm-water species, like tilapia. Additionally, the physical configuration of the incubation jars, combined with the controlled aeration system, can be easily adjusted to accommodate different egg buoyancies and biological characteristics, whether the eggs are pelagic (floating), demersal (sinking), or adhesive. As detailed by Gisbert et al. (2022) regarding phylogenetically related groups such as Siluriformes, the physical flow, aeration, and handling requirements vary significantly depending on distinct egg morphotypes; thus, the structural adaptability of our system provides a substantial experimental advantage for larviculture over traditional, single-purpose incubators. This adaptability highlights the technological potential of these devices to meet the diverse physicochemical requirements of various fish species, offering a highly functional tool for both experimental research and commercial hatcheries.
Acknowledgements. Fabricio Falcon (CICYTTP of Diamante, Entre Ríos, Argentina); to the Universidad Autónoma de Entre Ríos (UADER) for the training, support and mentoring provided to the team during the development of the work. This work received financial support from the Faculty of Science and Technology of the Autonomous University of Entre Ríos (Argentina), under Grant: Insertion Research and Development Project with resolution "CS" No. 175-23, and "Galápagos" Aquarium (Private producer).
Credit authorship contribution statement. MME: Conceptualization, Methodology, Research, Data curation, Formal analysis, Resources, Writing – original draft, Writing – review and editing. CLG: Research, Resources, Writing – original draft, Writing – review and editing, Supervision, Funding acquisition. LFM: Conceptualization, Methodology, Research, Data curation, Formal analysis. SMS: Resources, Writing – review and editing, Supervision. FAE: Conceptualization, Methodology, Resources, Writing – review and editing, Supervision, Fundraising.
Declaration of competing interest. The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability. Data openly available in a public repository that does not issue DOIs: The data that support the findings of this study are openly available in GiHub - "FedericoLoker/incufish" at https://github.com/FedeLoker/incufish.