Revista Veterinaria. 2026; 37(1)
https://doi.org/10.30972/vet.3719291
Artículo Original




1Laboratorio de Zoonosis y otras Enfermedades Transmitidas por Vector, Centro de Investigaciones Regionales "Dr. Hideyo Noguchi", Universidad Autónoma de Yucatán, Mérida, México. Avenida Itzaes, No. 490 x calle 59, Col. Centro, 97000, Mérida, Yucatán, México.
2Laboratorio de Enfermedades Emergentes y Reemergentes, Centro de Investigaciones Regionales "Dr. Hideyo Noguchi", Universidad Autónoma de Yucatán. Mérida, Yucatán, México. Avenida Itzaes, No. 490 x calle 59, Col. Centro, 97000, Mérida, Yucatán, México.
3Facultad de Ciencias Biológicas y Agropecuarias Región Poza Rica-Tuxpan, Universidad Veracruzana. Carretera Tuxpan Tampico Km 7.5, Universitaria, 92870 Alto Lucero, Tuxpan de Rodríguez Cano, Veracruz, México.
4Departamento de Salud Animal y Medicina Preventiva, Facultad de Medicina Veterinaria y Zootecnia, Universidad Autónoma de Yucatán. Carretera Mérida-X'matkuil Km 15.5, 97100 Mérida, Yucatán, México.
5Laboratorio de Diagnóstico Clínico en Materia Zoosanitaria, Facultad de Medicina Veterinaria y Zootecnia Campus II, Universidad Autónoma de Chiapas. Calz. Emiliano Zapata Km 8, Del Frigorífico, 29060, Tuxtla Gutiérrez, Chiapas, México.
c8 gabriela.flota@correo.uady.mx
gabriela.flota@correo.uady.mx
c11 antonio.torres@correo.uay.mx
antonio.torres@correo.uay.mx
Recibido: 09 diciembre 2025
Aceptado: 21 abril 2026
Abstract
Ehrlichia and Anaplasma are bacterial genera (family Anaplasmataceae, order Rickettsiales) that include tick-borne species responsible for emerging and reemerging diseases of importance to both animal and public health. Dogs living in regions with abundant tick vectors may act as hosts for Ehrlichia and Anaplasma species. The objective of this study was to determine the DNA detection frequency of Ehrlichia and Anaplasma, characterize the circulating species, and identify factors associated with their occurrence in dogs from a peri-urban community in Yucatan, Mexico. A total of 67 dogs (n=67) from Ucú, Yucatan, were included in the study. Blood samples were collected, and demographic and environmental characteristics were recorded. Genomic DNA was extracted and used for the detection of Ehrlichia and Anaplasma by endpoint PCR. In addition, bioinformatic tools and phylogenetic analyses were used to characterize the detectedspecies. Chi-square or Fisher's exact test, as appropriate, was performed to identify significant associations (p<0.05) between positive cases and the response variables. Overall, 62.6% (42/67) of dogs tested positive for Anaplasmataceae, 61.1% (41/67) for Ehrlichia, and 17.9% (12/67) for Anaplasma. Sequence and phylogenetic analyses revealed homologies with E. canis, A. platys, and A. phagocytophilum. Significant associations were found between Ehrlichia frequency and the variables "non-ideal" body condition (p=0.042), zone of the dwelling "interior" (p=0.009), and zone of the dwelling "exterior" (p=0.002), while the zone of the dwelling "central" (p=0.006) was associated with Anaplasma frequency. The circulation of these bacteria was confirmed in the studied dogs. These findings suggest that the distributions of E. canis and Anaplasma spp. is not homogeneous within the study area. Future studies should evaluate the eco-epidemiological factors influencing the local distribution of these pathogens.
Key words: Pets, canines, veterinary medicine, public health, tick-borne bacteria, ectoparasites.
[Pega aquí tu resumen en español]
Resumen. Ehrlichia y Anaplasma son géneros bacterianos (familia Anaplasmataceae, orden Rickettsiales) con especies transmitidas vectorialmente por garrapatas. Algunas especies causan enfermedades emergentes y reemergentes que son importantes para la salud pública y animal. Los perros de regiones con abundantes vectores pueden hospedar bacterias de Ehrlichia y Anaplasma. El objetivo fue reportar la frecuencia de ADN de Ehrlichia y Anaplasma, caracterizar las especies e identificar factores asociados con su frecuencia en perros de una comunidad periurbana de Yucatán, México. Se estudiaron 67 (n=67) perros de Ucú, Yucatán, de los cuales se recolectaron muestras de sangre y se obtuvieron las características demográficas y del sitio de estudio. Se extrajo el ADN genómico de las muestras y se utilizó para detectar Ehrlichia y Anaplasma mediante PCR punto final. Además, se emplearon herramientas bioinformáticas y árboles filogenéticos para caracterizar las especies de Ehrlichia y Anaplasma. Se realizó la prueba de c2 o la prueba exacta de Fisher, según corresponda, para identificar la asociación significativa (p<0,05) entre los casos positivos y las variables de respuesta. El 62,6% (42/67) de los perros fue positivo para Anaplasmataceae, el 61,1% (41/67) para Ehrlichia y el 17,9% (12/67) para Anaplasma. Las herramientas bioinformáticas y los árboles filogenéticos revelaron homologías con E. canis, A. platys y A. phagocytophilum. Las pruebas estadísticas mostraron asociación entre las variables condición corporal "no ideal" (p=0,042), zona de la vivienda "interior" (p=0,009) y zona de la vivienda "exterior" (p=0,002) con la frecuencia de Ehrlichia, y la variable zona de la vivienda "central" (p=0,006) con la frecuencia de Anaplasma. Se demostró la presencia de estas bacterias en los perros estudiados. Los resultados sugieren que la distribución de E. canis y Anaplasma spp. no es homogénea en el sitio de estudio. Estudios futuros deberían evaluar los factores ecoepidemiológicos que influyen en la distribución de la frecuencia de Ehrlichia y Anaplasma, al menos a nivel local.
Palabras clave: Mascotas, caninos, medicina veterinaria, salud pública, bacterias transmitidas por garrapatas, ectoparásitos.
Ehrlichia and Anaplasma are genera of bacteria in the family Anaplasmataceae. Both include species transmitted by hard ticks of the genera Amblyomma, Ixodes, and Rhipicephalus. These bacteria pose significant public health concerns due to their potential to infect humans and their negative impact on animal health, particularly in dogs and cats (Buysse et al. 2024). Furthermore, specific pathogen species infect livestock, including cattle and sheep, depending on the pathogen and the level of vector exposure (Dantas-Torres and Otranto 2017, Buysse et al. 2024).
Ehrlichiosis and Anaplasmosis are emerging tick-borne diseases and the second-most commonly diagnosed vector-borne diseases globally, after Lyme disease (Borreliosis). The primary species responsible for these diseases include E. chaffeensis, which causes Human Monocytic Ehrlichiosis and has been reported in Mexico (Franco-Zetina et al. 2022), and A. phagocytophilum, which causes Human Granulocytic Anaplasmosis and has been reported worldwide (Buysse et al. 2024). Additionally, E. canis and A. platys exhibit variable prevalence rates in dogs, particularly in tropical and subtropical regions, including the Yucatan Peninsula in southeastern Mexico (Ojeda-Chi et al. 2019a, Lira-Amaya et al. 2023). This region also has several identified arthropod vectors (Ojeda-Chi et al. 2019b, Dzul-Rosado et al. 2023).
Dogs serve as accidental hosts and reservoirs for various vector-borne pathogens (Arroyo-Ramírez et al. 2022). They also harbor ectoparasites, such as ticks and fleas, which transmit these pathogens (Cruz-Bacab et al. 2021). As a result, dogs play a crucial role in the epidemiology of certain vector-borne diseases, such as Ehrlichiosis and Anaplasmosis (Cermeño et al. 2025). Documented cases and varying prevalence rates of E. canis, A. platys, and A. phagocytophilum have been observed on both national and regional levels. However, a limited number of studies have investigated the factors influencing the frequency and distribution of these microorganisms in dogs, particularly focusing on intrinsic population characteristics (e.g., age, sex, vaccination status) (Ojeda-Chi et al. 2019a, Merino-Charrez et al. 2021). Therefore, this study aims to report the frequency of Ehrlichia and Anaplasma DNA in dogs from a peri-urban community in Yucatan, Mexico, and to identify factors associated with their frequency.
Type and site of study. A descriptive cross-sectional study was conducted on dogs from the community of Ucú, Yucatan, Mexico (89° 44' and 89° 51' meridians, and 20° 58' and 21° 10' parallels). The municipality covers an area of 192.89 km² and is located 7 m above sea level. The climate in most of the region is warm and semi-dry, with some areas of sub-humid conditions and summer rain. The annual temperature is 26 °C, with a maximum of 36 °C and a minimum of 16 °C. The dominant vegetation consists of low deciduous forest with transitional elements, including areas of cutting and forage pastures (Arroyo-Ramírez et al. 2025).
Study period and studied population. The study period was from August to October 2021. Due to COVID-19 health restrictions, only a limited number of dwellings were visited. These dwellings were selected for convenience, based on the consent of the occupants, who had been previously invited to participate in this study.
The spatial distribution model for the studied dwellings was developed using the methodology outlined by Ponce-Saavedra et al. (2019). In summary, two perpendicular lines were drawn along the town's main streets, extending outward from the town center to the outskirts, creating four quadrants. Next, three points were marked on each line to define concentric circles, establishing zones categorized as the interior, central, and exterior of the study site (Suárez-Galaz et al. 2024).
In addition to the signed informed consent form, inclusion in the study required the presence of dogs during the visit (only one visit per dwelling was conducted) and a signed permit authorizing work with the animals. Exclusion and elimination criteria included dogs under three months of age (as reported by the owner), owners who did not sign the informed consent form, dogs that exhibited aggression when handled, and dogs that had received antimicrobial treatment within three months before the visit (as reported by the owner).
Demographic data collection for the studied dogs. During dwelling visits, we collected data on the study dogs, reported by their owners or observed directly. 1. Ectoparasite infestation (ticks, fleas, or lice) regardless of the level of infestation, according to Torres-Castro et al. (2022). The categories included: negative (no) or positive (yes). Due to the study's aim, ectoparasites were not collected or identified; 2. Body condition: no ideal (emaciated, very thin, thin, overweight, heavy, obese or grossly obese) or ideal (physical findings: ribs are easily palpable with minimal fat covering; visual appearance: a visible waist behind the ribs is observed when viewed from above, and an abdominal tuck is evident when viewed from the side), according to the World Small Animal Veterinary Association Global Nutrition Committee guidelines; 3. Contact with other animals inside the dwelling (no or yes; other dogs, cats, and other domestic or wild animals); 4. Access to the outside of the dwelling (no or yes); 5. Zone of the dwelling where the dog lived (interior, central, or exterior).
Due to health restrictions imposed by the COVID-19 pandemic, which limited the duration of dwelling visits, the authors did not establish procedures, such as clinical records, to verify the data obtained from the owners. However, in cases where the dogs had access to veterinary services, vaccination and deworming records were obtained.
Collection and processing of blood samples. Previously, an experienced veterinarian immobilized the dogs. Three milliliters of whole blood were obtained from each dog studied by venipuncture of the cephalic or saphenous vein using a sterile syringe (BD Plastikpak®, USA). The samples were then placed into sterile tubes containing the anticoagulant EDTA (BD VacutainerTM, USA), which had been labeled with the animal's information (project identification). The samples were taken in the owner's presence, in accordance with the current regulations for handling and collection of animal samples in Mexico (NOM-062-ZOO-1999). Under field conditions, the samples were stored and transported in portable refrigerators (coolers) with refrigerants to the Emerging and Reemerging Diseases Laboratory at the “Dr. Hideyo Noguchi” Regional Research Center (CIR)-UADY for processing.
The transport time of the blood samples was 40 to 60 minutes from the study site to the laboratory for processing. This timeframe minimizes cell lysis and ensures sample stability. Likewise, during this time, the portable coolers remained closed and protected from direct sunlight to maintain a stable cold chain.
Detection of Ehrlichia and Anaplasma DNA. In the laboratory, blood samples were centrifuged at 3,500 rpm (2,000 g) at 24 °C for 10 minutes to separate white blood cells from the plasma, which were then transferred into sterile 1.8 ml tubes (Eppendorf®, Germany) and stored at -29 °C until further use.
Genomic DNA was extracted from both fractions using the Wizard® Genomic DNA Purification Kit (Promega®, USA) according to the manufacturer's instructions. The concentration (ng/mL) and purity of the extracted DNA were assessed using a Nanodrop-2000 (Thermo Scientific®, USA; Serial No. 8220). The samples were stored at ultra-low temperatures (-72 °C) until used in molecular reactions to detect Ehrlichia and Anaplasma.
A strategy was implemented first to detect Anaplasmataceae DNA, followed by detection of Ehrlichia and/or Anaplasma, and subsequently to identify the species using bioinformatics and phylogenetic analysis. Initially, it was used in a conventional PCR using the oligonucleotides EHR16SDF and EHR16SDR to amplify a conserved region of the 16S rRNA gene present in the Anaplasmataceae family (Parola et al. 2000). Samples that tested positive in this PCR were further analyzed using two specific molecular reactions targeting conserved fragments of the 16S rRNA gene in Ehrlichia (Chung et al. 2021) and Anaplasma (Silaghi et al. 2017).
The products obtained from all reactions were visualized on 2% agarose gels stained with ethidium bromide, along with the Invitrogen™ DNA Standard 1 kb Plus molecular weight marker (USA). The gels were visualized on the Bio-Rad® Gel Doc XR+ photo documentation system (USA; Serial No. 721BRO4265) to record the results.
Table 1 shows the oligonucleotides and reaction conditions (reagents, cycles, and temperatures) used to amplify DNA fragments from Anaplasmataceae, Ehrlichia, and Anaplasma in the blood samples of the dogs studied.

bp: base pairs; ’: minutes; ’’: seconds
Sequencing and identification of Ehrlichia and Anaplasma species. PCR-positive products for Ehrlichia and Anaplasma were sent to the Molecular Biology Laboratory at the “Salvador Zubirán” National Institute of Medical Sciences and Nutrition for Sanger sequencing. The obtained sequences (432-520 bp) were cleaned and aligned using the Clustal W paired-end algorithm in MEGA X. These sequences were then compared with homologous and reference sequences of valid species from the Ehrlichia and Anaplasma genera, which are stored in GenBank, using the BLAST-N tool with the Megablast algorithm (Felsenstein 1985).
Phylogenetic tree construction. Global alignments were performed for each bacterial genus using MEGA X with Clustal W. These included sequences generated from this study, as well as those obtained from the NCBI nucleotide sequence database (GenBank). After completing the alignments, phylogenetic trees were constructed using the maximum likelihood method (Tamura et al. 2004), with 1,000 bootstrap replicates in MEGA X. The best nucleotide substitution model was selected based on the lowest Bayesian Information Criterion (BIC) value (Felsenstein 1985).
Statistical analysis. The analysis of associated factors focused on the number of dogs that tested PCR-positive for Ehrlichia and Anaplasma. The remaining variables were considered independent. Separate analyses were performed for each zone (interior, central, and exterior). A 2 x 2 contingency table was created, and both c2 and Fisher's tests were applied using XLSTAT for Excel (version 2022.1). Results were considered significant at p≤0.05, with 95% confidence. Specifically, p-values were calculated using Fisher's exact test for variables with counts of ≤5 (Pértega-Díaz and Pita-Fernández 2004).
Bioethics Committee. The Bioethics Committee of the Campus de Ciencias Biológicas y Agropecuarias-Universidad Autónoma de Yucatán (UADY) approved this study (minute: CB/CCBA/M/2021/003).
Sixty-seven dogs were studied, of which 40.3% (27/67) were female, and 59.7% (40/67) were male. Most of the dogs were mixed breed (95.5%, 64/67) and adults (73.1%, 49/67). The majority of the dogs (92.5%, 62/67) had ectoparasite infestations, and 91.9% (57/62) had tick infestations. The most common body condition observed was ideal (53.7%, 36/67). Additionally, most dogs had contact with other animals (91%, 61/67) and had access to the outside of the dwelling (79.1%, 53/67). A greater number of dogs were studied in dwellings located in the exterior zone (38.8%, 26/67), followed by those in the central zone (31.3%, 21/67), and finally, dwellings in the interior zone (29.9%, 20/67).
Of the 67 dogs studied, 62.6% (42/67) tested positive for the Anaplasmataceae family; 61.1% (41/67) were positive for Ehrlichia, and 17.9% (12/67) tested positive for Anaplasma. Furthermore, co-occurrences of Ehrlichia and Anaplasma DNA were observed in 16.4% (11/67) of dogs.
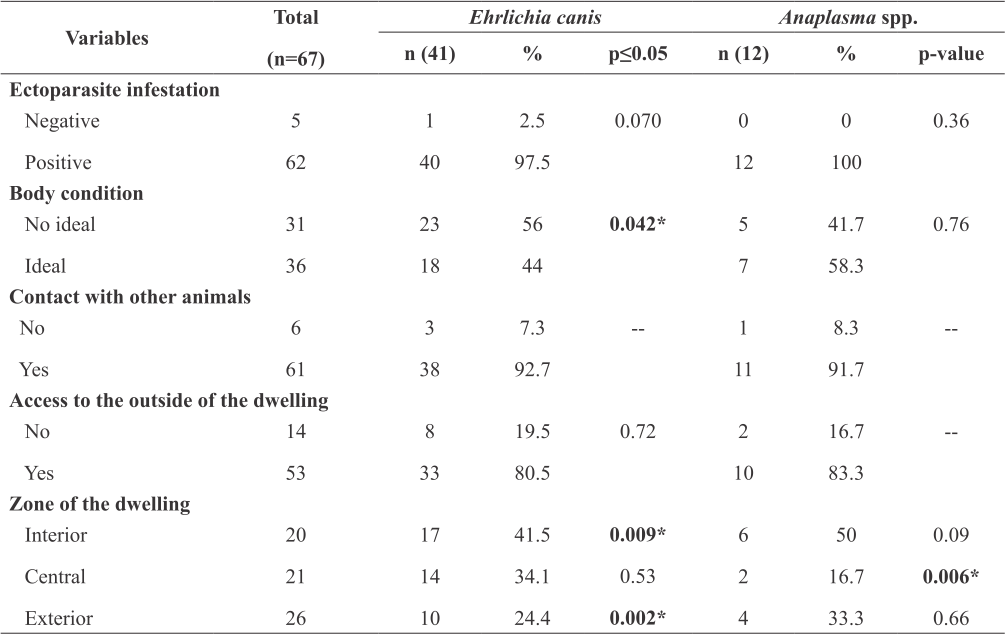
Of the Ehrlichia-positive dogs (n=41), almost all were ectoparasite-infested (ticks and fleas) (97.5%, 40/41). The most frequent condition was “no ideal” (56%, 23/41). Most had contact with other animals (92.7%, 38/41) and access to the outside of the dwelling (80.5%, 33/41). Regarding the dwelling zone, the positive dogs were distributed as follows: 17 (41.5%) in the interior, 14 (34.1%) in the central, and 10 (24.4%) in the exterior zone (Table 2).
Of the Anaplasma-positive dogs (n=12), all were infested (100%, 12/12). The most common condition was “ideal” (58.3%, 7/12); most dogs had contact with other animals (91.7%, 11/12) and had access to the outside of the dwelling (83.3%, 10/12). The dwelling zone was divided as follows: 50% (6/12) in the interior, 33.3% (4/12) in the exterior, and 16.7% (2/12) in the central zone (Table 2).
Statistical analysis revealed an association between Ehrlichia frequency and the independent variables: "non-ideal" body condition (p=0.042), the zone of the dwelling "interior" (p=0.009), and the zone of the dwelling "exterior" (p=0.002). Similarly, for Anaplasma frequency, the variable the zone of the dwelling “central” showed a significant association (p=0.006) (Table 2).
Alignment analysis with the recovered sequences of the Ehrlichia 16SrRNA gene fragment (GenBank accessions OR005546-54) showed 100% identity and homology with the E. canis MN227484.1 sequence. For Anaplasma, identity and coverage ranged from 99.79% to 100% (GenBank accession numbers OR006514-19) with the A. platys MK814414.1 sequence; A. phagocytophilum (GenBank accession numbers OR006521) obtained 100% identity and coverage with the CP046639.1 sequence.
*Statistically significant.
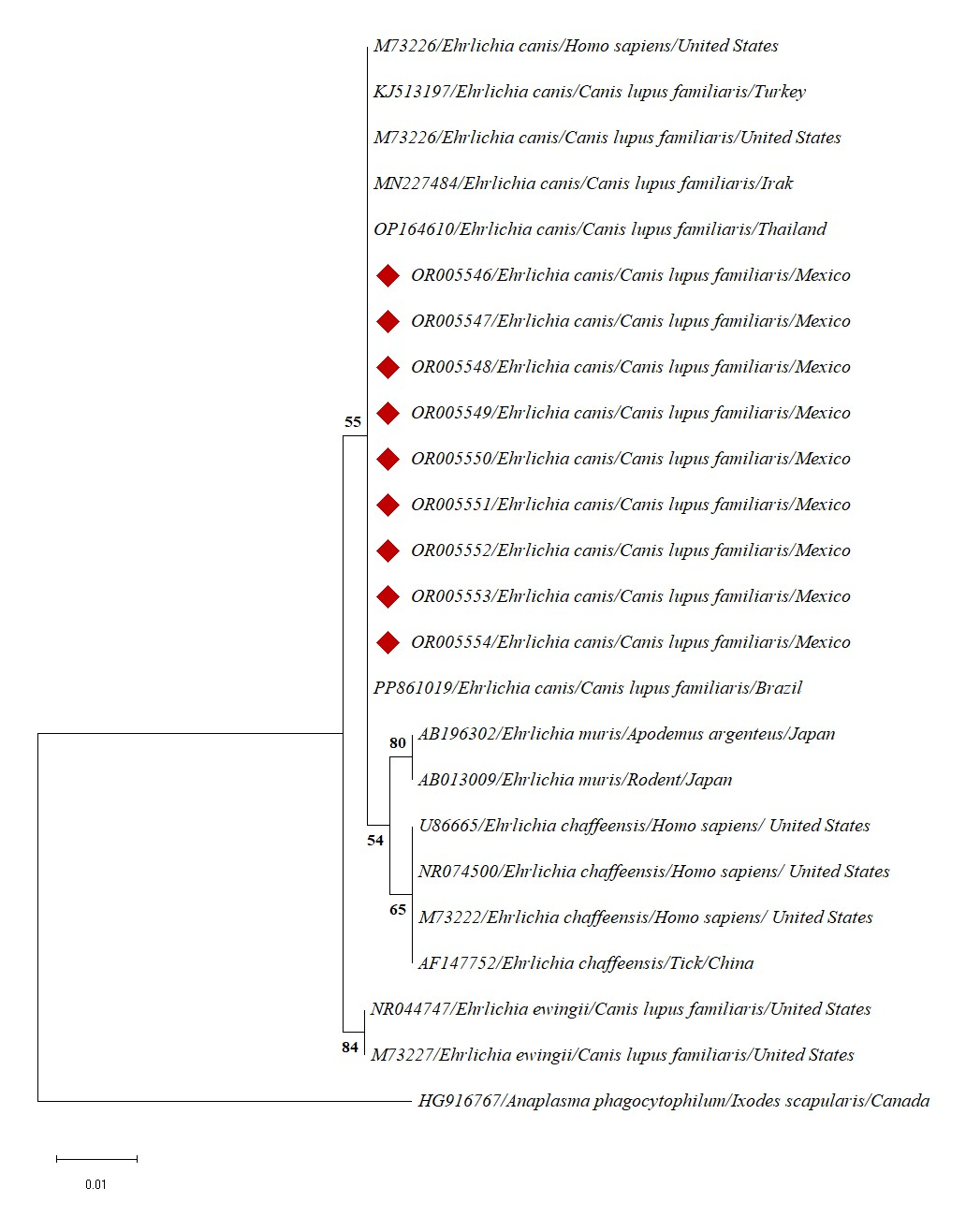
Phylogenetic trees complemented the information provided by bioinformatics analyses. In the case of Ehrlichia, all sequences formed a clade with E. canis sequences from several dogs from different countries (the United States, Turkey, Iraq, Thailand, and Brazil) and with one sequence from a human (Figure 1).
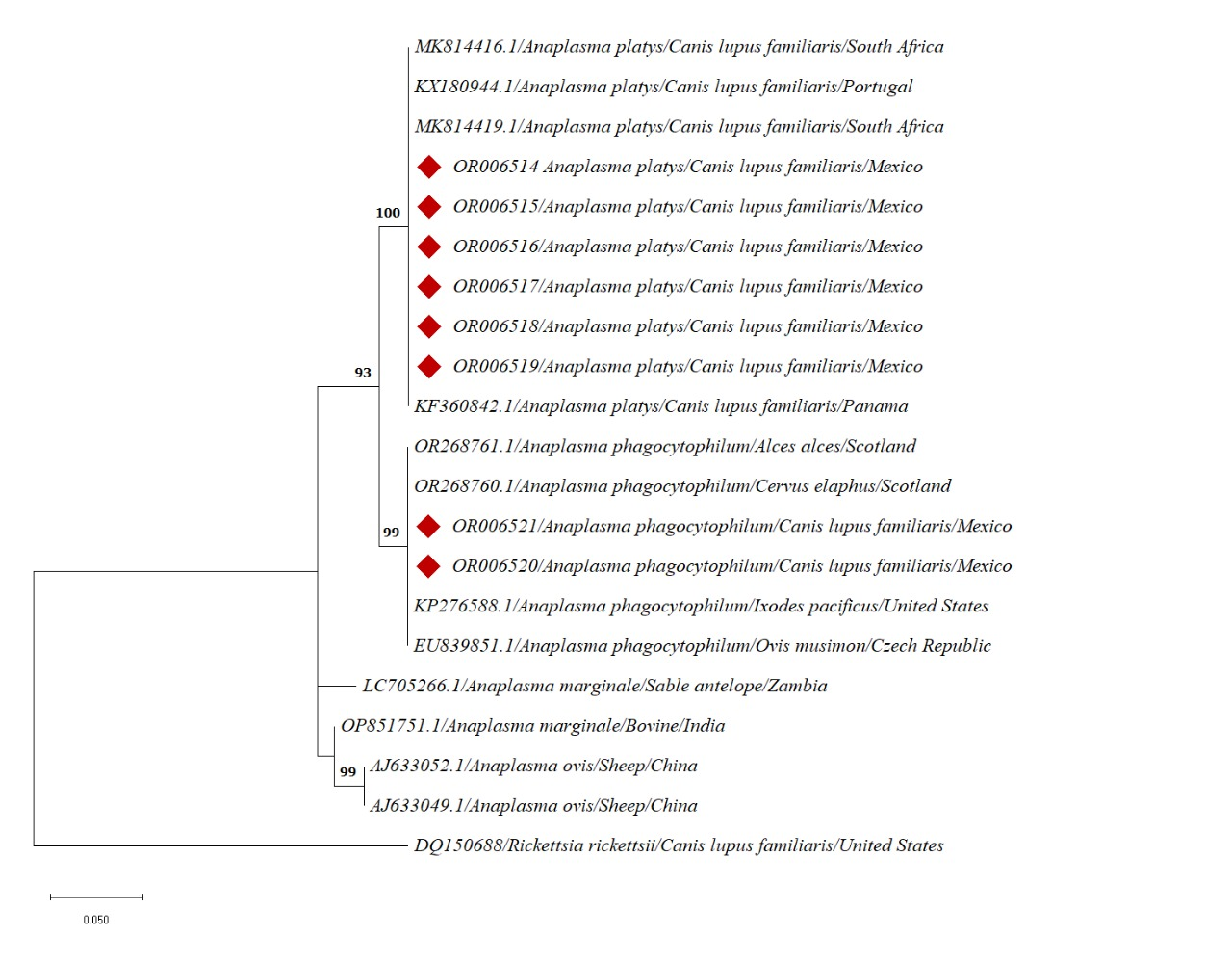
In the case of A. platys, the recovered sequences formed a clade with 100% branch support, with sequences obtained from dogs from South Africa, Portugal, and Panama. The A. phagocytophilum sequences formed a clade with 99% branch support, with sequences recovered from different hosts (Alces alces, Cervus elaphus, and Ovis musimon). This clade included a sequence retrieved from the tick Ixodes pacificus (Figure 2).
Dogs harbor numerous species of hard ticks described as vectors of Ehrlichia and Anaplasma species. They are therefore valuable elements in the epidemiological cycles of the diseases they cause (Solís-Hernández et al. 2018, Ojeda-Chi et al. 2019a,b, Little et al. 2021, Dzul-Rosado et al. 2023, Rodríguez-Vivas et al. 2024, 2025a). Likewise, the presence of infected dogs with any of these bacterial genera generates risks for the transmission of pathogens to humans and other susceptible hosts (Little et al. 2021, Balmori-de la Puente et al. 2024, Cermeño et al. 2025), because they eventually bring closer and maintain populations of ectoparasite vectors, mainly ticks and fleas, in anthropized sites such as dwellings, peridomiciles, vacant lots, parks, public markets, etc. (Ojeda-Chi et al. 2019b, Torres-Castro et al. 2020).
Infection with E. canis, A. platys, and A. phagocytophilum in the studied dogs may have public health implications for the study site (Cermeño et al. 2025). In Mexico, E. canis was identified in a person with close contact with dogs infected with the same bacteria (Silva et al. 2014). Otherwise, A. platys has been detected in owners of infected dogs in the United States (Breitschwerdt et al. 2014). Anaplasma phagocytophilum has significant zoonotic importance because it causes Human Granulocytic Anaplasmosis in countries across all continents (Dantas-Torres and Otranto 2017), including Mexico (Sosa-Gutiérrez et al. 2021).
Records of dogs coinfected with pathogens transmitted by ectoparasites, especially E. canis and Anaplasma spp., are frequent (Little et al. 2021, Balmori-de la Puente et al. 2024). At the national level, Almazán et al. (2016) reported the presence of Anaplasma spp. and E. canis in dogs from the “La Comarca Lagunera”. Similarly, Rojero-Vázquez et al. (2017) in dogs from Campeche, Aragón-López et al. (2021) in dogs from Sonora, and López-Valencia et al. (2024) in dogs from Baja California. At the international level, records of coinfection in dogs have been reported in the USA (Bowman et al. 2009), Peru (Carbajal-Ruiz and Vilela-Velarde 2024), Colombia (Cermeño et al. 2025), and Venezuela (Cermeño et al. 2025), among other countries and territories. These findings collectively reflect the wide geographical distribution of E. canis and Anaplasma spp. This is due, among other epidemiological factors, to the geographical distribution and abundance of their competent vectors (Dantas-Torres and Otranto 2017, Rodríguez-Vivas et al. 2023).
According to Rodríguez-Vivas et al. (2025b), serological evidence (generated through ELISA and IFI) against E. canis has been documented in dogs from 24 states in Mexico, including Yucatan, with seroprevalences ranging from 0.9% (State of Mexico) (Bedoya et al. 2023) to 100% (Chihuahua) (Lira-Amaya et al. 2023), due to adaptation to different anthropized environments and the geographical distribution and high abundance in Mexico of the primary vector of E. canis, R. sanguineus s.l. In contrast, records with molecular tools (PCR and multiplex PCR) are fewer. They are distributed across nine states, including Yucatan, with frequencies ranging from 4% (Coahuila [Almazán et al. 2016] and Durango ;Almazán et al. 2016;) up to 69.2% (Yucatan) (Díaz-Medina et al. 2016).
The frequency of Ehrlichia in the studied dogs was 61.1% (41/67). Compared with reports from Mexico, it is only lower than the 69.2% identified by Díaz-Medina et al. (2016) in dogs from Yucatan. In dogs from Chihuahua, Rodríguez-Alarcón et al. (2020) reported a similar frequency of 61.3%. In Yucatan, Ojeda-Chi et al. (2019a) identified R. sanguineus s.l. as the primary biological vector of E. canis, with a detection frequency of 7.7%. Other ticks collected from dogs in Yucatan with evidence of E. canis belong to the genus Amblyomma, including A. ovale (Ojeda-Chi et al. 2019b).
A homologous sequence of A. platys was identified in R. sanguineus s. l. infesting dogs in a rural community in Yucatan, which shares sociodemographic characteristics similar to those of the study site (Dzul-Rosado et al. 2023). This tick is recognized as the primary vector of this bacterium in dogs worldwide (Cardona-Arias et al. 2019, Chao et al. 2024). However, to the authors' knowledge, this is the first record of A. platys in dogs from southeastern Mexico.
At the national level, there is evidence of A. platys in dogs from various states, including Sonora (Aragón-López et al. 2021), Tamaulipas (García-Mora et al. 2024), Veracruz (Jerez-Sulvarán et al. 2024), and the region known as “La Comarca Lagunera” (Almazán et al. 2016). Internationally, A. platys has been reported in dogs from Egypt (Selim et al. 2021) and Grenada (Alhassan et al. 2021), among other countries. These records, as with E. canis, reflect the wide geographical distribution of this bacterium worldwide and its primary vector (R. sanguineus s. l.) (Cardona-Arias et al. 2019).
The primary vectors of A. phagocytophilum are ticks of the genus Ixodes, which are associated with enzootic and zoonotic transmission cycles (El Hamiani Khatat et al. 2021). However, it has also been identified in naturally infected R. sanguineus s. l. (Escárcega-Ávila et al. 2018, Balmori-de la Puente et al. 2024). Several Ixodes species parasitize dogs from different locations in Yucatan (Ojeda-Chi et al. 2019b, Rodríguez-Vivas et al. 2024), indicating the presence of eco-epidemiological factors that facilitate the transmission of this bacterium in the region. However, this is the first report of A. phagocytophilum in dogs from Yucatan, Mexico. At the national level, Rojero-Vázquez et al. (2017) identified A. phagocytophilum in dogs from Campeche at a frequency of 17%, and Escárcega-Ávila et al. (2018) identified it in dogs from Chihuahua at a frequency of 13%. In dogs from Tamaulipas, Merino-Charrez et al. (2021) found no evidence of Anaplasma spp.
Internationally, records of A. phagocytophilum have been reported in dogs from all continents, with infection rates up to 57.3% (Hamidinejat et al. 2019).
This study also highlights that the dwelling's geographic location (zone) is a factor associated with dogs infected with Ehrlichia and Anaplasma. This suggests that the distribution of these pathogens among dogs at the study site is not uniform, indicating that ecological and epidemiological factors, which were not identified in this study, influence their distribution, at least on a local scale. However, given the cross-sectional design and non-probability sampling, it is important to note that associations do not imply causality; the results are only exploratory at the local level.
In the case of the "interior zone," the result may be related to the greater number of dogs roaming in this zone of the study site (field observation), which increases direct contact between dogs or infested ticks areas (Saleh et al. 2019), generating higher transmission rates through the exchange of ectoparasite vectors compared to dogs from other zones of the study site (Huerto-Medina et al. 2015). Rhipicephalus sanguineus s. l. is recognized for its high population densities in dogs from tropical and subtropical regions of Mexico, particularly in regions such as the Yucatan Peninsula (Ojeda-Chi et al. 2019b, Sánchez-Montes et al. 2021, Rodríguez-Vivas et al. 2023). Additionally, a study conducted in Baja California found that interactions with other dogs increase the risk of exposure to vector-borne diseases, such as Ehrlichiosis (López-Valencia et al. 2024).
Another factor that may influence the frequency of Ehrlichia is the higher density of dwellings in the “interior zone” (as observed in the field). Rhipicephalus sanguineus s. l. is known to be endophilic, meaning it is highly adapted to environments inhabited by humans, such as the interior of homes, peridomiciles, gardens, and kennels. This adaptation contributes to higher abundances and densities of this tick (Rodríguez-Vivas et al. 2023). Further epidemiological studies are necessary to understand how dog population density and dwelling conditions (density, distribution, distance, etc.) influence the prevalence of E. canis in the study region.
The “exterior zone” was also associated with the frequency of Ehrlichia in the dogs studied. According to the owners, dogs living in these dwellings have contact with nearby forest patches and other mammals that host ectoparasites (Rojero-Vázquez et al. 2017), increasing the risk of Ehrlichia transmission (López-Valencia et al. 2024). Solís-Hernández et al. (2018) noted that dogs with access to forested or wild environments are more likely to be infested with various tick species. Additionally, several researchers have observed a higher prevalence of tick-borne diseases in dogs that frequent forested areas, attributing this to their continuous exposure to vectors (Cermeño et al. 2025).
Moreover, while the difference in the number of dogs between the different zones is not statistically significant, the higher number of dogs in dwellings of the exterior zone may have influenced the frequency of Ehrlichia. Greater canine population density is directly related to the prevalence and infestation of vector ticks. Accordingly, an increased number of hosts raises the likelihood of tick feeding and facilitates rapid reproduction (Cermeño et al. 2025).
It is unclear why dogs from dwellings in the "central zone" were associated with Anaplasma frequency. However, it is known that Ehrlichia and Anaplasma coinfection in the same dog population is due to the presence and circulation of ectoparasites that host both bacterial genera. It is worth noting that dogs in this area exhibited a high rate of infestation, which has been associated with Anaplasma infection in a previous study by Selim et al. (2021).
The distribution of E. canis and Anaplasma spp. is heterogeneous across the study site, and there are significant associations between dwelling location (zones) and infection frequency. This information can be used to develop action programs in the identified high-risk areas. These programs could include campaigns for tick and flea control, health and sanitary education, public awareness events, and promoting responsible pet ownership, particularly in the interior and exterior areas for Ehrlichia and the central zone for Anaplasma spp.
Regarding the information provided by bioinformatics analysis and genetic trees, a high homology (100% for A. platys and 99% for A. phagocytophilum) was observed between Anaplasma sequences obtained from the studied dogs (A. platys: OR006514-19; A. phagocytophilum: OR006520, OR006521) and sequences obtained from dogs and other hosts in other countries of the world (for A. platys: South Africa [MK815516.1, MK814419.1] and Portugal [KX180944.1]; for A. phagocytophilum: Scotland [OR268761.1, 0R268760.1], the USA [KP276588.1], and Czech Republic [EU839851.1]). This is due, among other factors, to the high degree of conservation of the amplified fragment of 16SrRNA of the genus Anaplasma (Kolo 2023).
Although molecular identification of Anaplasma spp. is important, obtaining isolated sequences does not necessarily imply active or established transmission in the region. For this, it is necessary to evaluate the presence, abundance, and distribution of Ixodes tick species and R. sanguineus s. l., as well as their capacity to act as vectors of A. platys and A. phagocytophilum.
In the case of Ehrlichia, the sequences recovered from the studied dogs (OR005546-54) formed a clade with sequences retrieved from dogs from Turkey (KJ513197), the USA (M73226), Iraq (MN227484), Thailand (OP164610), and Brazil (PP861019). This clade included a sequence obtained from a human from the United States (M73226). The value of the branch support (55%) is due to the high heterogeneity of the amplified fragment of the 16SrRNA gene. Similar results are reported by Rodríguez-Rojas et al. (2024), which includes E. canis sequences recovered from dog ticks in Nuevo León, Mexico (not included in the corresponding tree).
This study has limitations (cross-sectional design, convenience sampling during the COVID-19 pandemic, limited collection and identification of ectoparasites, and lack of confounder adjustment) that should be considered when interpreting the findings, which are specific to the sample studied and not necessarily generalizable to the entire region due to acknowledged sampling bias.
Future research should employ longitudinal designs with probabilistic sampling, integrate entomological sampling by dwelling and microhabitat characterization, apply multivariate models, and expand the number of molecular markers to improve taxonomic resolution and, potentially, guide cost-effective interventions by area. Practical campaigns for homeowners on safe tick inspection and removal, as well as coordination between veterinary and local medical surveillance, will strengthen early detection and mitigation of zoonotic risk.
Acknowledgments. To all those who allowed us into their homes despite the health restrictions of the COVID-19 pandemic. To Dr. Rosa María Galaz for her support during the fieldwork. The National Council of Science and Technology, now the Secretariat of Science, Humanities, Technology, and Innovation (SECIHTI) of Mexico, awarded a master's scholarship to Osorio-Primo, T.
CRediT authorship contribution statement. O-P, T.: Conceptualization, formal analysis, investigation, methodology, validation, visualization, writing – original draft. L-C, C.: Conceptualization, data curation, formal analysis, funding acquisition, methodology, project administration, resources, supervision, validation, writing – original draft. N-P, H., S-G, A.R., A-R, A.: Formal analysis, methodology, validation, software. S-M, S., R-V, R.I., B-G, M.: Conceptualization, methodology, validation. F-B, G., O-V, J.L.: Investigation, methodology. T-C, M.: Conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, project administration, resources, supervision, validation, writing – original draft, writing – review & editing. All authors reviewed the original draft.
Declaration of competing interest. The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability. Data will be made available on request.
.